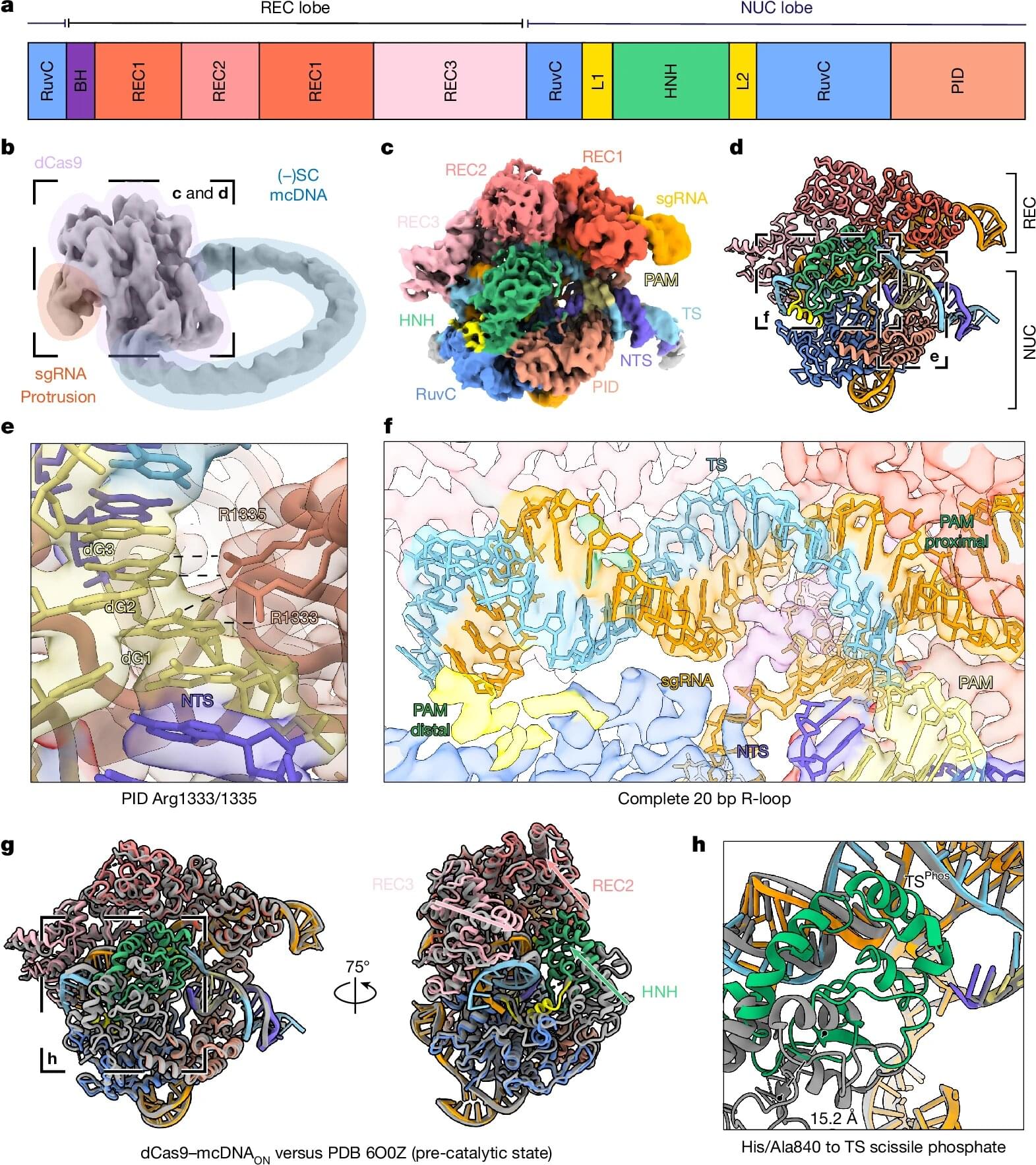
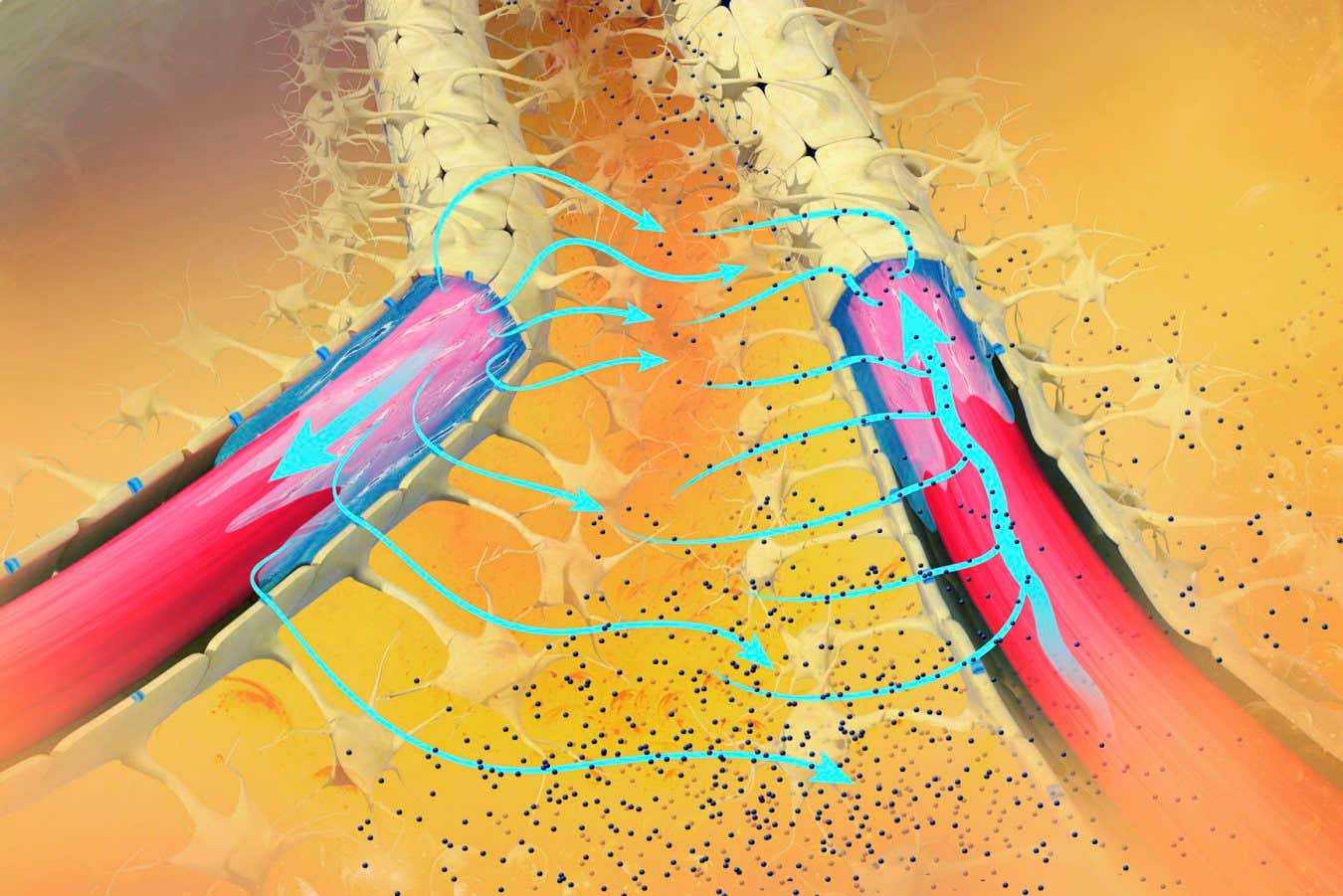
CRISPR is a powerful DNA-editing tool that has underpinned huge advancements in human health care in the last decade. It is a precision tool, but is not perfect, and misplaced DNA edits can compromise safety and efficacy, costing billions each year. Researchers at the MRC Laboratory of Medical Sciences (LMS), Imperial College London and the University of Sheffield have published research in Nature showing that the physical twisting of DNA plays an important role in these mistakes. Using a newly developed platform of tiny (nanometer-sized) DNA circles, called DNA minicircles, the team captured never-before-seen interactions between CRISPR and DNA, providing insights that could help eradicate errors altogether.
CRISPR-Cas9 has transformed biology by giving scientists a programmable way to cut and edit DNA. Its ever-growing impact includes groundbreaking therapies for genetic diseases such as sickle cell anemia and an increasing role in personalized cancer treatment and rapid diagnostics. But even carefully designed CRISPR systems can sometimes cut DNA sequences that were not the intended targets.
“It’s a tool that is not perfect and can introduce errors and make edits where it shouldn’t make them,” says Professor David Rueda, head of the Single Molecule Imaging group at the LMS and Chair in Molecular and Cellular Biophysics at Imperial College London. “And it’s an important problem for the industry. It’s been estimated to be $0.3 to $0.9 billions per year in industry costs, in profiling off-targets, redesigning guides and delays.”