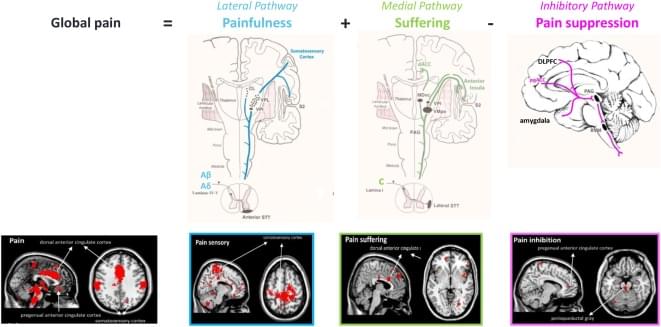
Pain is an unpleasant sensory and emotional experience associated with actual or potential tissue damage. Chronic pain, with a prevalence of 20–30% is the major cause of human suffering worldwide, because effective, specific and safe therapies have yet to be developed. It is unevenly distributed among sexes, with women experiencing more pain and suffering. Chronic pain can be anatomically and phenomenologically dissected into three separable but interacting pathways, a lateral ‘painfulness’ pathway, a medial ‘suffering’ pathway and a descending pain inhibitory pathway. One may have pain(fullness) without suffering and suffering without pain(fullness). Pain sensation leads to suffering via a cognitive, emotional and autonomic processing, and is expressed as anger, fear, frustration, anxiety and depression.