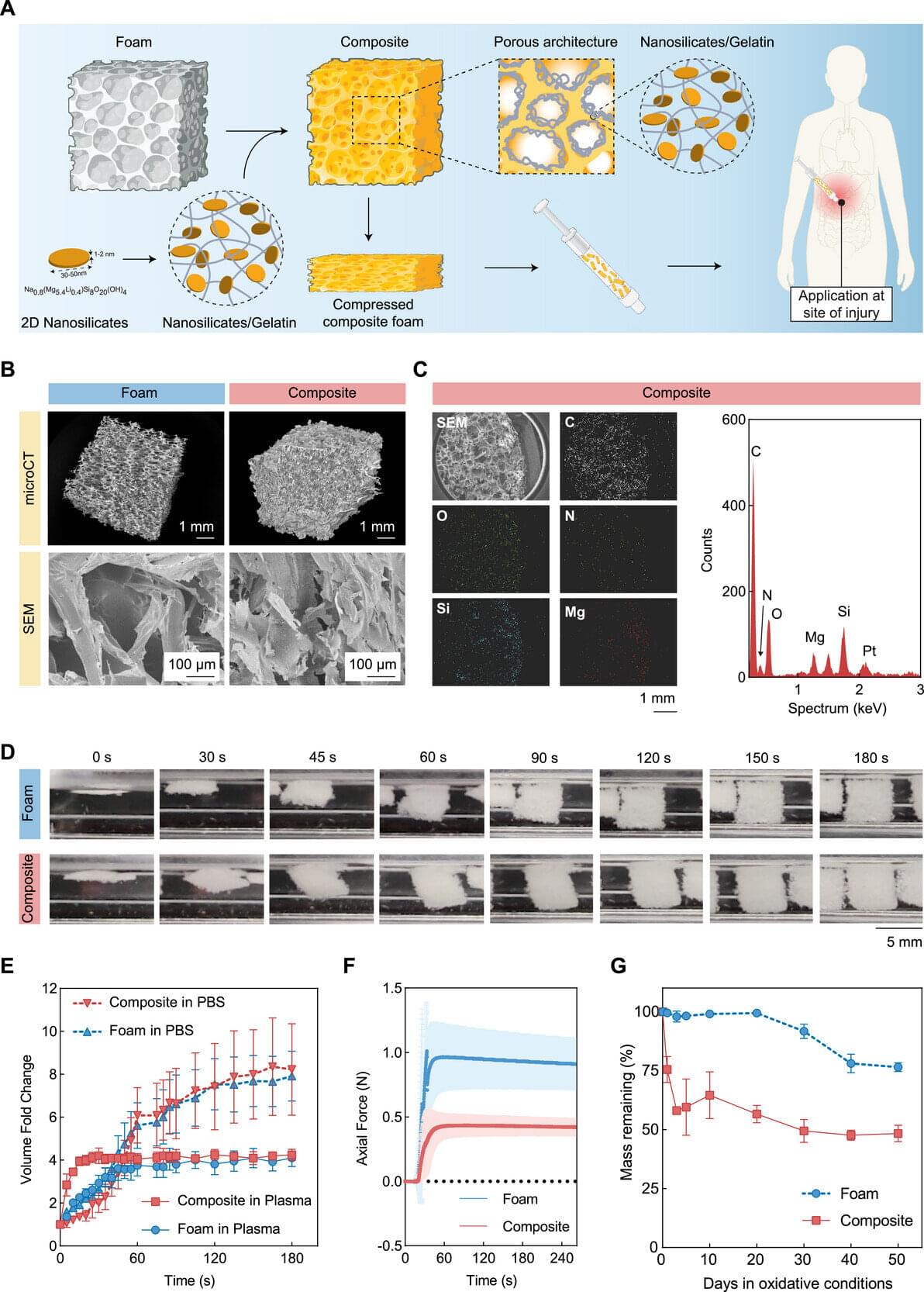
Traumatic injury is the third leading cause of death in the state of Texas, surpassing strokes, Alzheimer’s disease and diabetes, according to the Centers for Disease Control and Prevention. A massive number of these deaths are the result of uncontrolled bleeding. “Severe blood loss can rapidly lead to hemorrhagic shock,” said Dr. Akhilesh Gaharwar, a biomedical engineering professor at Texas A&M University. “Many patients die within one to two hours of injury. This critical period is often referred to as the ‘golden hour.’”
Gaharwar and his fellow researchers in the biomedical engineering department have found a way to extend this golden hour—using clay.
Gaharwar, Dr. Duncan Maitland and Dr. Taylor Ware are developing a suite of injectable hemostatic bandages —biomedical materials that stop bleeding and promote blood to clot faster. Their research is specifically targeting deep internal bleeding where traditional methods like compression are not possible.