C12 introduced a patented nanoassembly technology that enables precise carbon nanotube placement for future quantum processors.
Category: nanotechnology
Ultrathin nanotubes reach 1 nanometer, opening path to smaller electronics
Researchers in Japan have created some of the world’s smallest semiconducting nanotubes, structures 100,000 times thinner than a human hair. By growing molybdenum disulfide inside protective tubes of boron nitride, the researchers, including those from the University of Tokyo, produced highly uniform tubes just 1 nanometer wide, a scale at which it’s difficult to make stable nanotube structures. The work confirms decades-old theoretical predictions about how these ultrafine materials behave and could also provide a new route toward miniaturized electronic devices.
The research is published in the journal Science.
A few years ago, carbon nanotubes were attracting a lot of press attention. But there’s a new contender in the ring, and it offers some advantages over its carbon counterpart that could tempt engineers to design products around it.
Nano Weapons: The Invisible Machines Changing Future Wars!
In this video, we explore the incredible and terrifying world of nano-weapons — microscopic machines designed for the battlefields of the future. From invisible drones to molecular-level assassins, nanotechnology is revolutionizing modern warfare in ways the world has never seen before. Discover how these tiny machines can spy, sabotage, and even kill at the atomic scale. We’ll uncover real-world research, secret military projects, and the ethical dangers behind the next generation of warfare. The rise of nano-weapons could change the balance of global power forever — but are we ready for what’s coming? Watch till the end to understand the full potential and risks of these microscopic war machines.
Hashtags (12):
#NanoWeapons #FutureWarfare #Nanotechnology #MilitaryTech #WarInnovation #ScienceAndTechnology #NanoMachines #InvisibleWar #BattlefieldOfTomorrow #DefenseTechnology #AIWarfare #FutureScience.
Keywords (23):
nano weapons, microscopic machines, future warfare, military technology, nanotechnology, nano drones, nano robots, molecular weapons, defense innovation, secret military research, nano science, nano warfare, nano army, advanced technology, invisible battlefield, modern weapons, AI warfare, nanobots, nano defense, future military, scientific discovery, advanced warfare, nano.
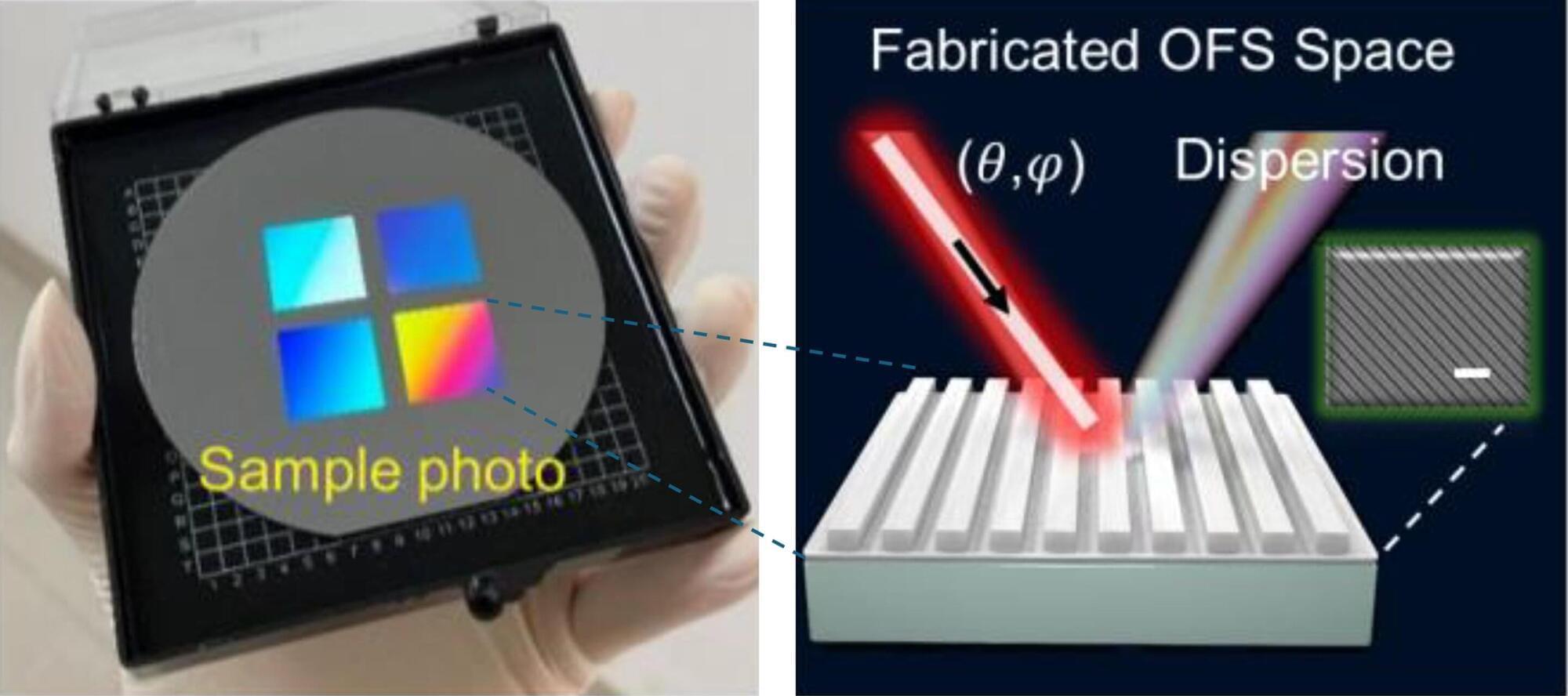
Teaching AI to design optical surfaces using real-world imperfections
Designing surfaces that precisely control how light behaves at the nanoscale is tricky. Optical Fourier surfaces, which are nanostructured gratings that redistribute light into specific directions and wavelengths, hold enormous potential for compact spectrometers, augmented-reality displays, and advanced sensors. However, their standard design process relies on computer simulations that assume idealized conditions such as single-angle illumination and the absence of fabrication imperfections—a far cry from reality.
The gap between what simulations predict and what fabricated devices actually do has long frustrated researchers. It widens further when designers try to exploit one of the most powerful but underused design parameters: the angle of incoming light. Changing the incident angle can activate or suppress optical modes without any physical modification to the structure, effectively enabling multiple functions on a single device.
“This effectively introduces an additional degree of freedom beyond geometry, expanding the design space significantly,” said Associate Professor Dong Zhaogang from the Singapore University of Technology and Design (SUTD). “But its practical use has been limited because simulations at oblique incidence are often computationally unstable and costly, while real experimental systems involve angular distributions rather than single-incident angles.”
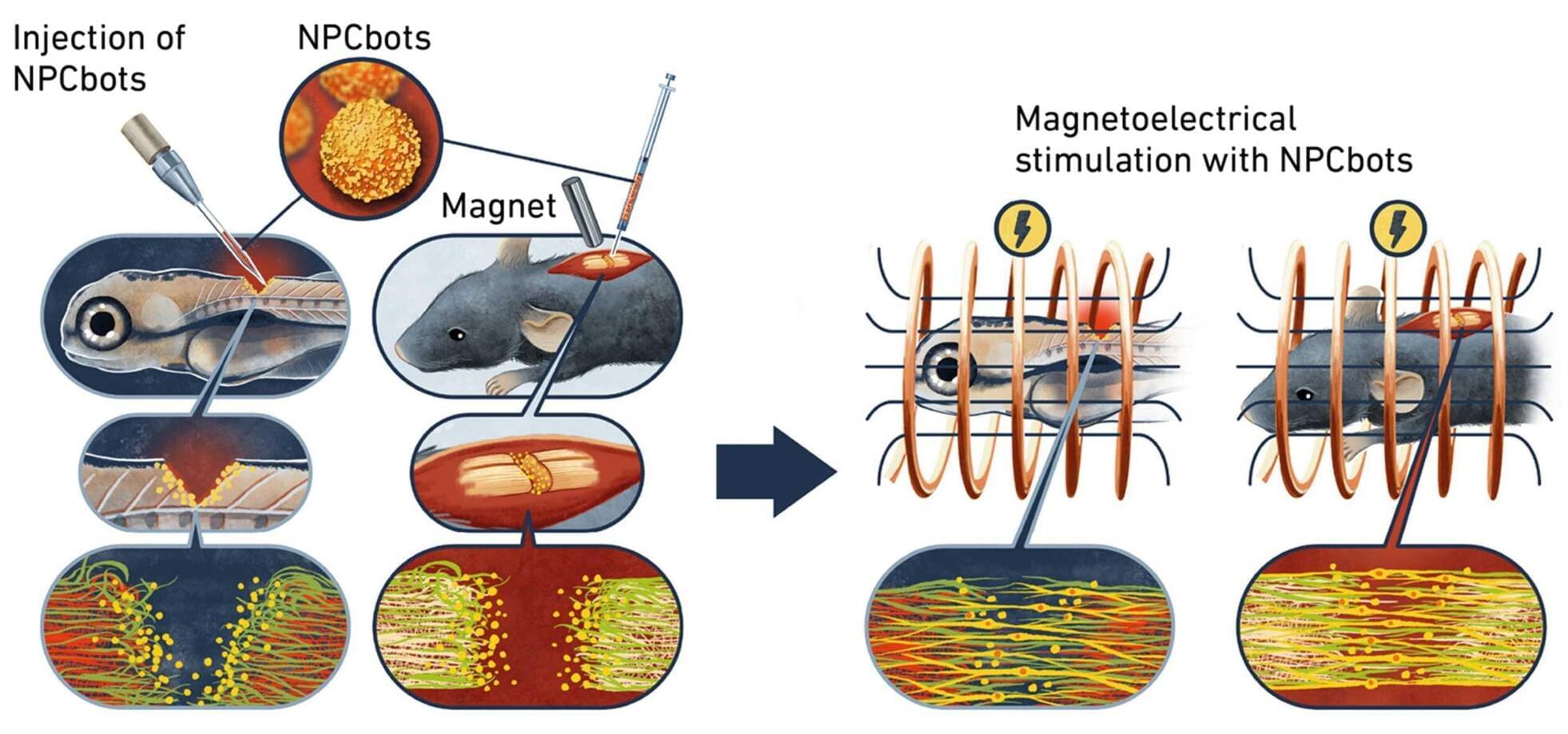
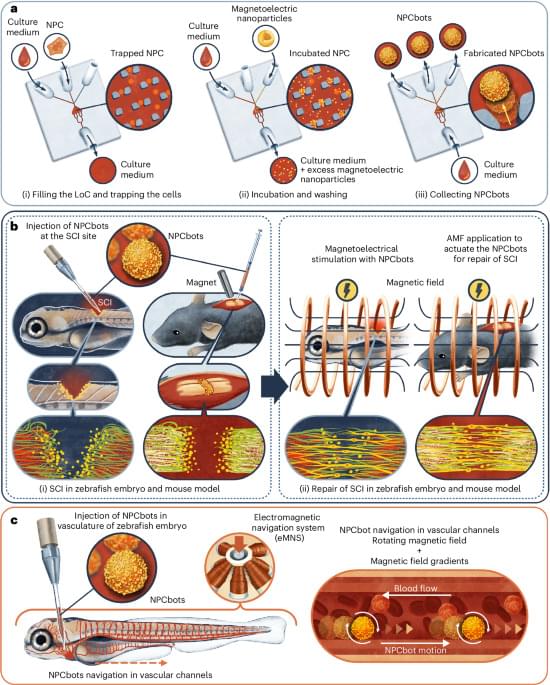
Biohybrid microrobots repair spinal cord by combining stem cells with magnetoelectric nanoparticles
Spinal cord injuries can have devastating consequences for those affected. Nerve cells in the spinal cord rarely regenerate naturally, while scarring often prevents the regrowth of nerve fibers. Modern therapies attempt to influence implanted stem cells using electrical stimulation to promote the growth of new nerve cells. This approach has several drawbacks: it requires implanted electrodes, and the transplanted cells do not always survive or integrate properly into the existing tissue.
Researchers in Zurich are pursuing a new approach, which they have published in the journal Nature Materials. This involves combining therapeutic stem cells with magnetoelectric nanoparticles in such a way that the cells can be guided magnetically to the precise site of an injury and stimulate the stem cells to accelerate repair.
To achieve this, the researchers created a biohybrid microrobot, which combines living neural progenitor cells (NPCs) with a technical component in the form of specially engineered nanoparticles.
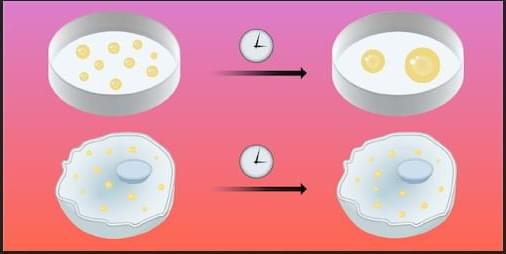
Why Nanoscale Droplets Don’t Coalesce
A well-shaken mixture of oil and vinegar will separate as the oil droplets eventually coalesce. Droplet growth, or coarsening, usually evolves according to standard rules. But puzzling exceptions persist. When two polymers are mixed in water and the concentration is high enough, droplets containing one or both species form and can remain stable for hours or days. These loose molecular condensates otherwise behave like liquid droplets, and they abound in biological cells. Now Feipeng Chen of the University of Hong Kong and his colleagues have developed a predictive model for coarsening behavior that works across a range of droplet sizes and explains why coarsening may be suppressed in living systems [1].
The researchers derived their model from observations of a solution containing water and two different polymers, opposite in charge and having very different molecular chain lengths. Using light-scattering techniques, the researchers monitored condensate growth over 12 hours. The initial size and subsequent growth rate of the liquid-like droplets, rich in both polymers, turned out to depend on the solution’s overall initial concentration. In the most dilute solutions, condensates tens-of-nanometers in diameter formed and promptly stopped growing for the remaining 12-hour observation period. In solutions having slightly higher concentration, hundreds-of-nanometer condensates formed and remained stable, then underwent abrupt, rapid growth in the later stages. And in the most concentrated solutions, micrometer-scale condensates formed and grew according to a power-law model.
Applying an electric field to the solutions indicated that the nanoscale condensates had significant surface charge. Modeling these measurements revealed that the asymmetric chain lengths of oppositely charged polymers imparted a net charge to the droplet surfaces. These charges led to size-dependent electrostatic barriers that drastically reduced merging efficiency below a critical diameter. The finding offers a principle for controlling size stability in biology, nanotechnology, and soft-matter assembly.
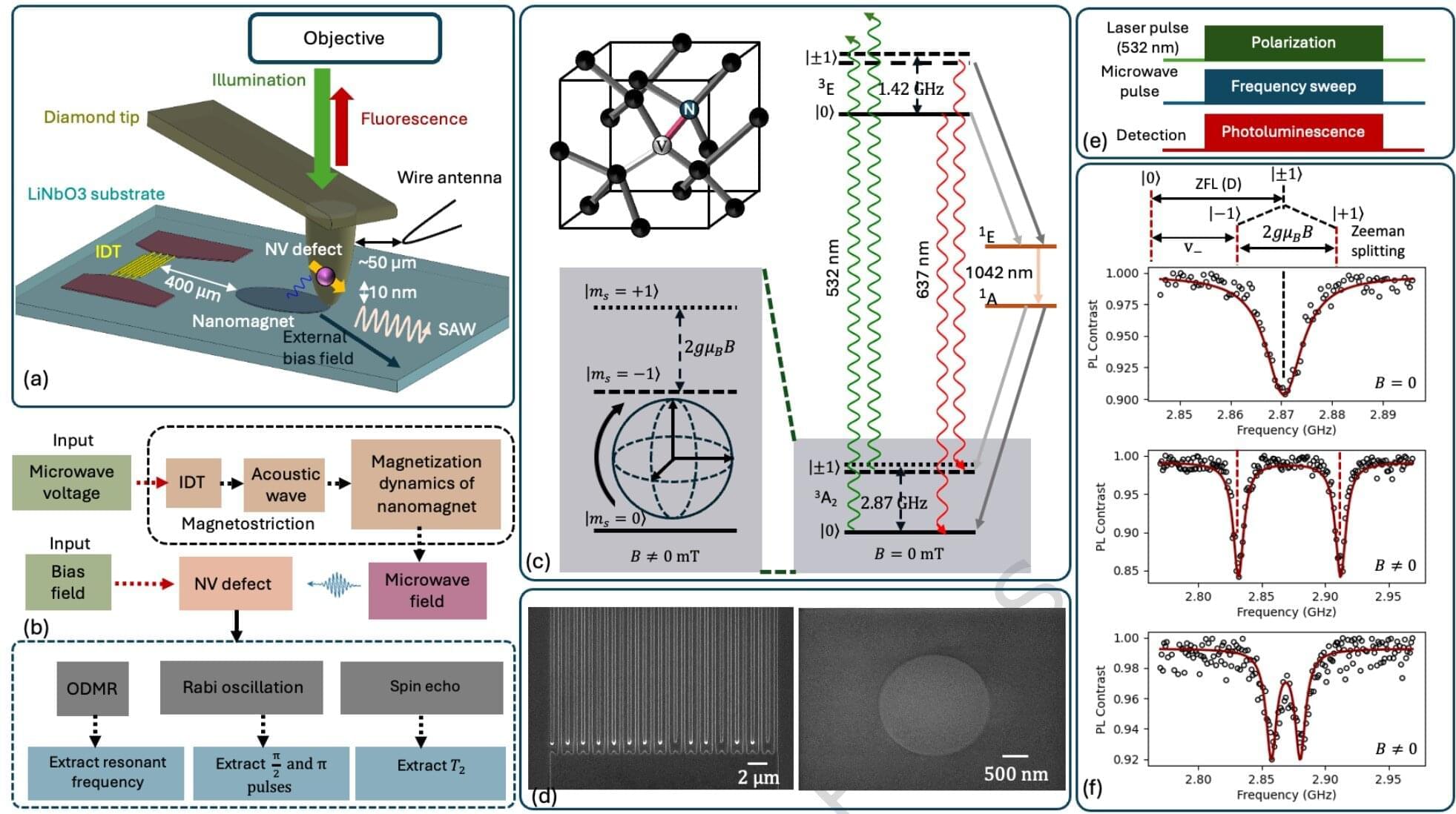
Nanomagnets control diamond qubits, pointing to more scalable quantum hardware
Quantum computing, once only a theoretical possibility, promises to deliver faster, more energy-efficient computers—but only if scientists can build and scale the hardware needed to run the machines. New research from Virginia Commonwealth University brings scientists one small step closer to quantum computing at a practical scale, which could help dramatically reduce energy usage and computing times in some industries.
In the study, recently published in Nature Communications, the researchers used minuscule magnets—twice as small as the wavelength of light—to create the building blocks of quantum computing, pioneering a technique that could decrease the physical space needed to create a viable quantum computer.
“This work has the potential to advance quantum computing,” said Jayasimha Atulasimha, Ph.D., a professor of mechanical and nuclear engineering in VCU’s College of Engineering and the study’s principal investigator. “We’re solving a specific problem for spin-based quantum computing, which has the potential for scaling.”
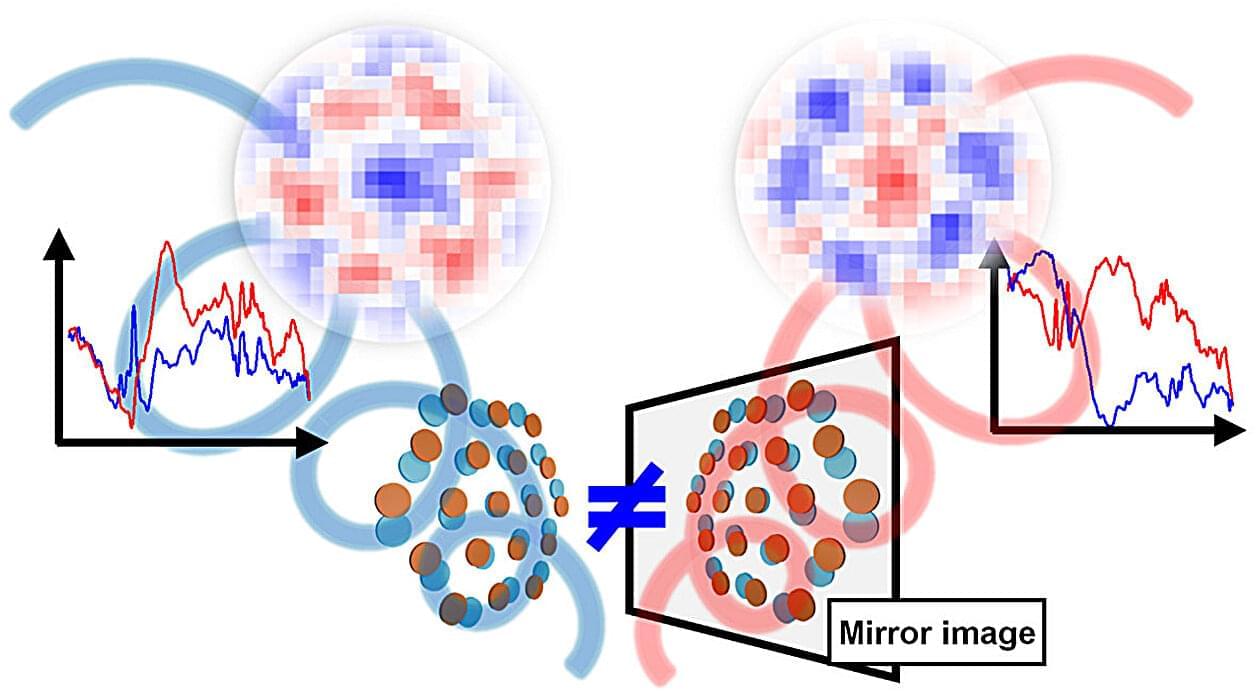
Terahertz imaging maps spatial chirality in materials with 100-micrometer resolution
In nature, there exist structures that are mirror images of each other but cannot be perfectly superimposed. These are known as chiral objects, derived from the Greek word for “hand,” since left and right hands share the same relationship. Although similar in structure, chiral molecules exhibit different behaviors, and chirality is central to life itself. DNA has a twisted chiral structure, and living organisms prefer one handedness over the other. This distinction is equally important in drug design, materials science, and nanotechnology.
One way to distinguish chiral molecules is by measuring their response to circularly polarized light in the terahertz (THz) region. THz waves lie between microwaves and infrared light and are especially sensitive to subtle collective motions and twisting structures in materials. However, conventional THz measurements average the signal across an entire sample, making it impossible to determine how chirality varies across different locations.
Now, researchers from Chiba University, Japan, and Tohoku University, Japan, have shown that this limitation can be overcome, allowing chirality to be visualized as two-dimensional images, much like creating a map of chirality across a material. The work appears in ACS Photonics.

Atomic reshuffle leads to record-breaking catalysts for hydrogen production
Researchers have discovered that atoms can be mixed, separated, and recombined within the same experiment, providing a pathway to a record-breaking catalyst for green hydrogen production. In their study, the team created nanoscale particles containing only a few dozen platinum and nickel atoms and observed unusual dynamic behavior in direct space and in real time. As the two metals separate from one another while maintaining an interface, they become highly active for electrochemical water splitting, leading to efficient hydrogen evolution.
The project was led by the University of Nottingham in collaboration with the University of Birmingham, Diamond Light Source, and Ulm University in Germany. The study appears in Advanced Materials.
Research team leader Dr. Jesum Alves Fernandes, from the School of Chemistry, University of Nottingham, said, “What makes this discovery exciting is that we can reversibly tune the structure of the particle while directly observing the process at the atomic scale. This opens a new strategy for designing adaptive catalysts for a wide range of applications.”