Startups are combining AI and genetics to make more food for less money


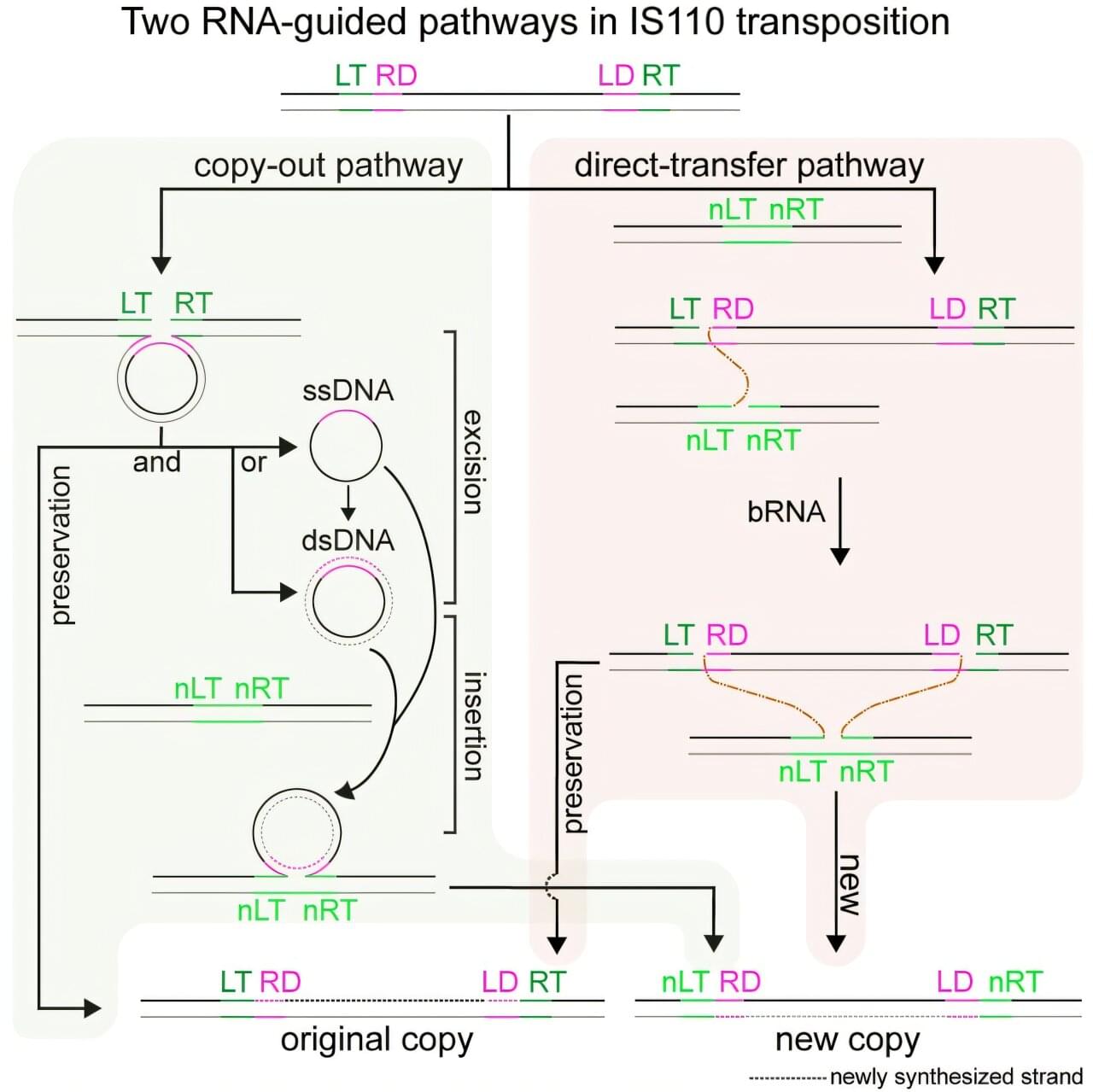
IS110 transposons are a large, diverse family of bacterial insertion sequences (IS elements)—small, mobile DNA elements that can move from one genomic location to another. They have recently attracted broad interest due to the finding that some of these transposons use a bridge RNA (bRNA) to recognize both donor DNA and target DNA.
Upon this discovery, researchers hoped that bRNA-guided transposon systems could offer a genome-editing strategy distinct from classical CRISPR-Cas nucleases and thereby enable programmable DNA integration. However, it remained unclear how IS110 elements insert donor DNA into target sites and whether these elements rely on one or multiple reaction pathways.
Now, a new study led by Xue Chaoyou from the Tianjin Institute of Industrial Biotechnology of the Chinese Academy of Sciences, in collaboration with Lou Huiqiang at China Agricultural University and RAO Shuquan from the Chinese Academy of Medical Sciences, answers these questions by showing that RNA-guided IS110 transposons use two mechanistically distinct pathways to mobilize DNA.



Gov. Jared Polis on Thursday declared a statewide drought emergency, citing the record-low snowpack and prolonged warmer temperatures across Colorado.
He also activated the next phase of the state’s drought response plan. Polis had placed Colorado under Phase 2 in March.
“Today, I am issuing a statewide drought emergency to support Coloradans, our economy, farmers and ranchers, and outdoor enthusiasts in the face of one of the most severe droughts in Colorado’s recorded history. With every county in the state experiencing drought conditions, activating Phase 3 of our Drought Response Plan allows us to better coordinate agencies, prepare for worsening conditions, and support Colorado communities, agriculture, water users, and our environment,” he said in a statement. “State agencies will do their part to reduce water usage at state facilities and I encourage every Coloradan to use water wisely.”

I’ve spent years watching finance and technology slowly adapt to one another, but the shift we’re looking at right now is going to change the entire landscape overnight. We need to stop thinking of AI as just a software tool or a cool shortcut for writing emails. We are officially entering an era where computational power is a foundational global commodity—and the standard unit of that commodity is the AI token.
Think of it like digital energy. Just as factories consume kilowatt-hours of electricity, modern enterprises now have to “burn” tokens to power their workflows. In my latest piece, I break down the massive hidden risk of letting a few Big Tech hyperscalers control both the production of this raw material and the infrastructure of exchange. This is where the banking sector has to step in, not just to cut their own costs, but to act as the ultimate market makers for artificial thought.
I dive deep into how banks will soon offer token futures markets—allowing companies to hedge their computing costs the exact same way airlines hedge aviation fuel—and how autonomous AI agents will soon be transacting with each other using tokenized value. The institutions that build these financial rails now will own the next century of commerce, while the rest risk being left behind in an aging system.
Click through to read the full breakdown on how the machine-to-machine economy is actually going to work!
(https://www.linkedin.com/pulse/new-gold-standard-when-ai-tok…Resilience over Political Influence: History shows that attempting to lobby a system to be “less exploitative” rarely works because the system is designed for extraction. True survival in this model might mean finding “off-grid” pockets where the resource demand is low enough to fly under the AI’s radar, or where the land is unsuitable for massive data centers.
I have spent a significant portion of my career watching the tectonic plates of finance and technology grind against each other. Usually, it is a slow, methodical process—a gradual shifting of legacy systems adapting to new digital realities. But every so often, a shift occurs that is so profound, it completely redefines the landscape overnight. We are standing on the precipice of one of those shifts right now.

Birgitta Schultze-Bernhardt and her team at the Institute of Experimental Physics at Graz University of Technology (TU Graz) have developed a new type of UV dual-comb spectrometer that detects gaseous air pollutants with unrivaled accuracy and sensitivity. Using ultraviolet double laser light, the device measures the concentration of harmful gases such as formaldehyde within half a second.
Thanks to its compact design and a measuring range of up to two and a half kilometers, the spectrometer is not only suitable for laboratory analyses, but also for mobile measurements in cities, industrial areas and agricultural regions.
The work is published in the journal PhotoniX.

Laser systems operating in the 2-micrometer wavelength range open diverse opportunities in medical technology, agriculture, and plastics processing. In the Eurostars project DECOMP, Laser Zentrum Hannover e. V. (LZH) has developed novel fiber optic components that overcome previous technical barriers.
Thulium-doped fiber lasers operate at a wavelength of approximately 2 micrometers, making them particularly well-suited for applications where conventional lasers reach their limits. However, commercially available laser sources that simultaneously offer high beam quality, sufficient laser power, and the necessary reliability in quasi-continuous-wave operation at power levels around 1 kilowatt have been lacking.
In the project, LZH scientists developed novel fiber optic components based on triple-clad fibers that enable a reliable and low-maintenance laser architecture. For the implementation of the final laser system, LZH collaborated with Futonics Laser GmbH as well as South Korean partners COSET, inc. and the Korean Photonics Technology Institute.

/ @lostpagesofscience.
Discover the incredible future of biohybrid robots, the revolutionary fusion of synthetic biology, artificial intelligence, and robotics! In this episode, we explore robots powered by living tissues, capable of self-repair, adaptation, and natural movements. Find out how these bioengineered robots can transform medicine, agriculture, environmental science, and prosthetics. Learn about the ethical considerations, safety challenges, and futuristic possibilities of combining biological materials with robotic systems.
🔔 Don’t forget to subscribe for cutting-edge science & technology content!
📌 Chapters:
00:00 — Introduction to Biohybrid Robots.
01:30 — What Are Biohybrid Robots?
03:50 — How Scientists Build Biohybrid Robots.
06:20 — Medical Applications & Healthcare.
08:45 — Revolutionary Prosthetics.
11:00 — Environmental Biohybrid Robotics.
13:20 — Agricultural Applications.
14:50 — Ethical & Safety Considerations.
17:00 — Future Possibilities.
19:00 — Conclusion & Call to Action.
SEO Keywords:
Biohybrid robots.
Synthetic biology and robotics.
Living robots.