The energy-efficient desalination system produces fresh water without chemical additives and transforms leftover salts into useful materials.
Category: sustainability

Aerosols may warm or cool the climate depending on timing, new study finds
A new study from the Hebrew University of Jerusalem challenges a long-held assumption in climate science by showing that aerosols—tiny particles suspended in the atmosphere—can either warm or cool the climate, depending on the time scale considered.
Led by Prof. Guy Dagan of the Fredy and Nadine Herrmann Institute of Earth Sciences, the research reveals that aerosol-cloud interactions can produce opposite climate effects in the short and long term. The findings, published in Nature Communications, offer a new explanation for why aerosols remain one of the largest sources of uncertainty in climate projections.
Aerosols come from a variety of natural and human-made sources, including air pollution, wildfires, sea spray and dust. Scientists have long known that these particles influence how clouds form and how much heat Earth retains, but accurately estimating their overall impact on climate has proved difficult.
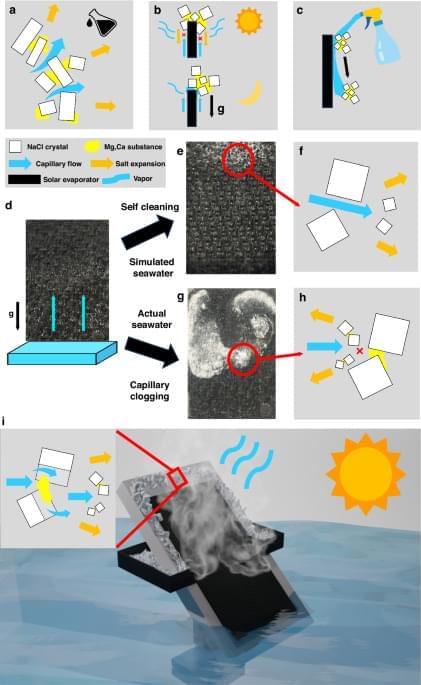
Additive-free and brine-discharge-free solar-thermal desalination with simultaneous complete mineral mining from ocean water
Tang, L., Singh, S.C., Wei, R. et al. Light Sci Appl 15, 246 (2026). https://doi.org/10.1038/s41377-026-02315-4
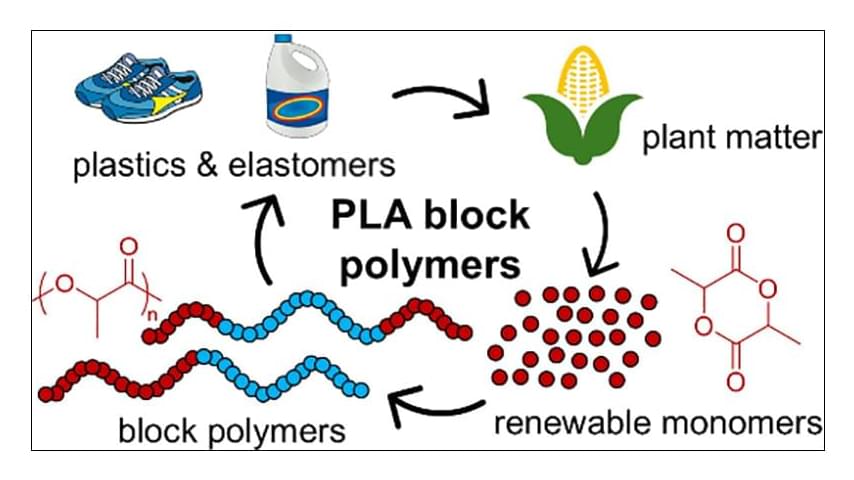
PLA Block Polymers: Versatile Materials for a Sustainable Future
Block polymers present an almost endless realm of possibilities to develop functional materials for myriad applications. The established self-assembly of block polymers allows researchers to access properties that are inaccessible in homopolymers. However, there is a need to develop more sustainable options than the current commodity block polymers. Derived from renewable resources and industrially compostable, poly(lactide) (PLA) is at the forefront of technological advancements in sustainable block polymers. Its material properties including high stiffness, relatively high glass transition temperature, and semicrystallinity in isotactic versions lend themselves to many applications, and its ease of synthesis provides a well-established platform for developing high-performance materials. This Perspective highlights recent advancements associated with PLA-containing block polymers, including their syntheses, mesostructural considerations, and mechanical properties, from resilient elastomers to tough plastics. We also give our perspective on the subfield of PLA block polymers, our outlook on the future, and our assessment of exciting developments yet to come.

Hidden meltwater found deep in Antarctic coastal waters reveals stronger climate impacts
Freshwater from melting Antarctic glaciers may be influencing the Southern Ocean in ways scientists have largely overlooked. New research, published in Frontiers in Marine Science, has found that glacial meltwater is not confined to the ocean’s surface, as previously assumed, but can also be detected much deeper in coastal waters along the Western Antarctic Peninsula.
The findings suggest that meltwater from glaciers is being transported and stored tens of meters below the surface, where it could alter ocean circulation, affect the movement of heat and nutrients, and influence how the region responds to climate change.
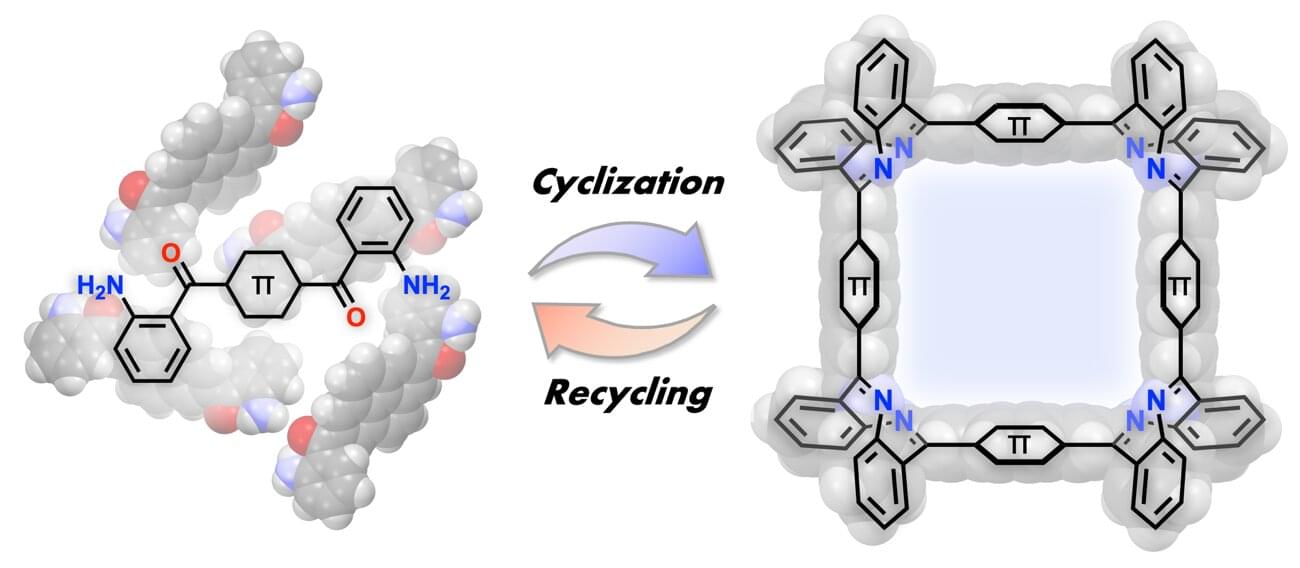
A new strategy for assembling π-conjugated panels into square molecules revealed
A research group has developed a new method for selectively synthesizing three-dimensional macrocycles,⁽¹⁾ in which four panels are arranged in a square, by connecting planar π-conjugated molecules⁽²⁾ at right angles.
This method is applicable to a wide variety of π-conjugated molecules and allows the size of the internal cavity to be designed. Furthermore, the resulting square macrocycles exhibit acid responsiveness, reversibly changing color under the action of a mild acid, while acid-mediated hydrolysis enables the starting monomers to be recovered in high yield—realizing a sustainable molecular synthesis that reverts to and regenerates the starting materials. The originality of this work lies in having a single imine bond play three roles: creating the shape, responding to stimuli and reverting back.
These research results were published in the Journal of the American Chemical Society on Monday, June 1, 2026. The team includes Associate Professor Yasutomo Segawa and Assistant Professor Takashi Harimoto at the Institute for Molecular Science (National Institutes of Natural Sciences) and the Graduate University for Advanced Studies (SOKENDAI).

Light pulses uncover Higgs mode that reshapes perovskite crystal symmetry
Waves of light and sound interact to drive electronic and structural changes in a perovskite crystal. At the atomic scale, nothing is ever truly still. Materials that appear perfectly rigid and motionless to the naked eye are in fact swarms of vibrating atoms. This motion is generally random and uncoordinated, but with the right input, the atoms in certain materials will start to move together, vibrating in sync.
These collective vibrations are a form of sound called phonons, and when tuned just right, they can influence a material’s structure and behavior in dramatic and useful ways. Researchers are working to understand and control this effect to optimize material properties and even access hidden phases of matter.
Scientists at the U.S. Department of Energy’s (DOE) Argonne National Laboratory are using light to drive phonon activity in a class of materials called metal halide perovskites, whose customizable structures and photosensitivity hold promise for use in next-generation solar cells, advanced sensors and quantum information technologies.

North Atlantic spring storms have grown more common since 1940, analysis reveals
Storm Dave, which swept across northern Europe over the Easter weekend, is an example of what new research from the University of Gothenburg has revealed. Spring storms forming over the North Atlantic have become more common than they were 80 years ago, and this is due to climate change.
In the Northern Hemisphere, storm seasons follow a seasonal cycle. Storms are weakest and least frequent in summer and most intense in winter. As a result of global warming, storm patterns and their course have changed, and several studies have indicated that winter storms appear to be occurring more frequently and with even greater intensity.

Glowing Blue Spider Among the Dozens of New Species Discovered in One Area on Research Expedition
A recent expedition to Central Africa has uncovered dozens of new species.
In February, a team of 16 specialists from Africa and around the world visited the Lisima plateau in eastern Angola and conducted a biodiversity survey, through which they discovered dozens of species unknown to science, according to The Wilderness Project, which led the survey.
The organization dedicated to studying and protecting Africa’s freshwater wilderness announced the findings from the remote scientific expedition in the area — seen as one of Africa’s last great biodiversity blank spots — in a news release obtained by PEOPLE on Wednesday, June 3.
