This website uses a security service to protect against malicious bots. This page is displayed while the website verifies you are not a bot.

Analysis of US mortality data from 1990 to 2023 indicates breast cancer, lung cancer, and leukemia deaths decreased in people younger than 50, while ColorectalCancer mortality increased, rising from fifth to first among cancer deaths in this age group.
The majority of cases present at an advanced stage, highlighting the need for increased symptom awareness and earlier screening.
National trends suggest ongoing prevention and early detection strategies are vital for addressing early-onset colorectal cancer.
This study examines changes in cancer mortality in the US for the 5 leading cancer-related deaths among people younger than 50 years over the past 3 decades.
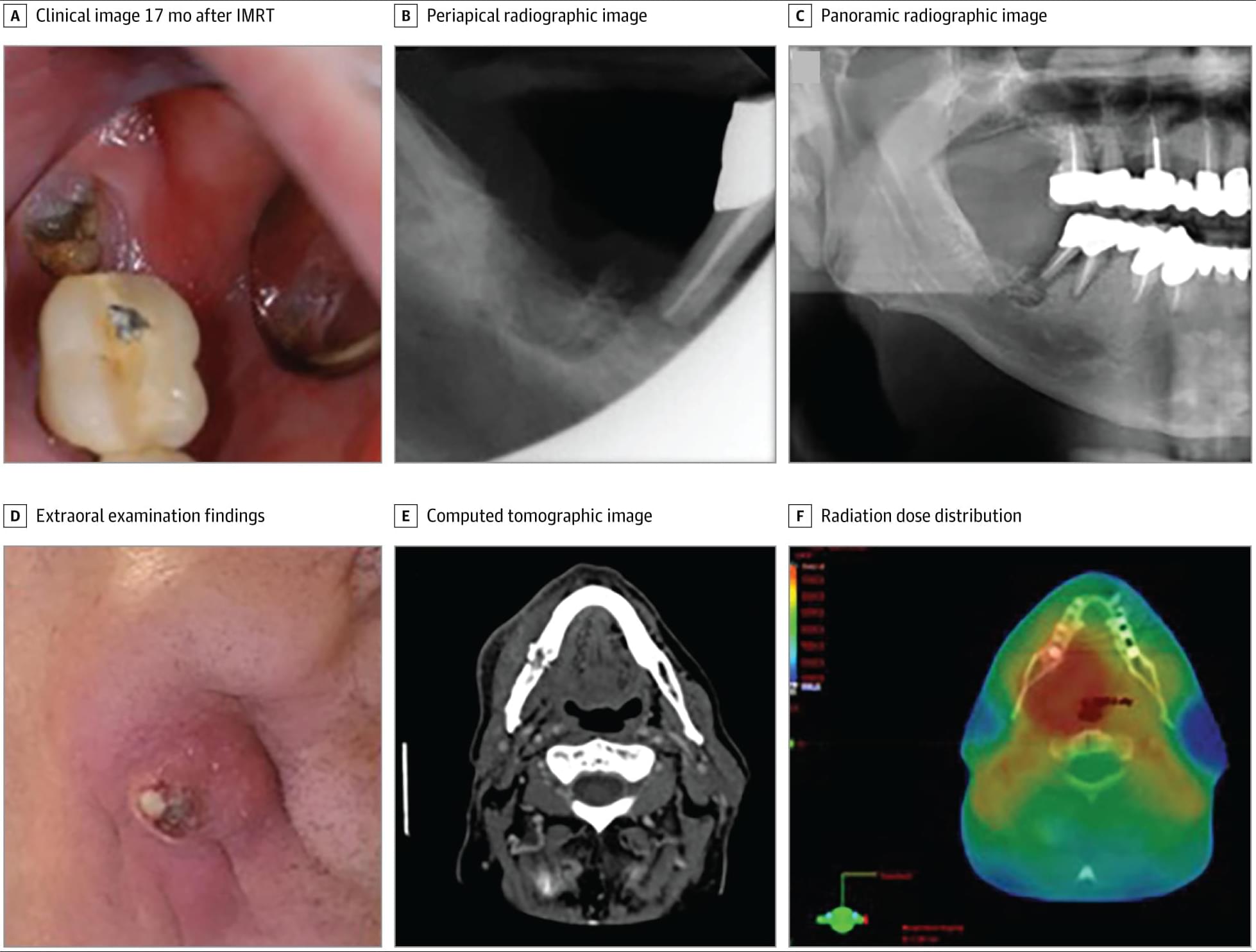
Among patients with oropharyngeal squamous cell #carcinoma, proton therapy for head and neck cancer was associated with a higher 3-year incidence of osteoradionecrosis compared with intensity-modulated radiation therapy.
Severe osteoradionecrosis rates were low and did not differ by modality.
This cohort study characterizes the incidence, severity, and predictors of osteoradionecrosis in patients with oropharyngeal squamous cell carcinoma treated with curative-intent radiotherapy and compares outcomes between those receiving proton therapy vs intensity-modulated radiation therapy.
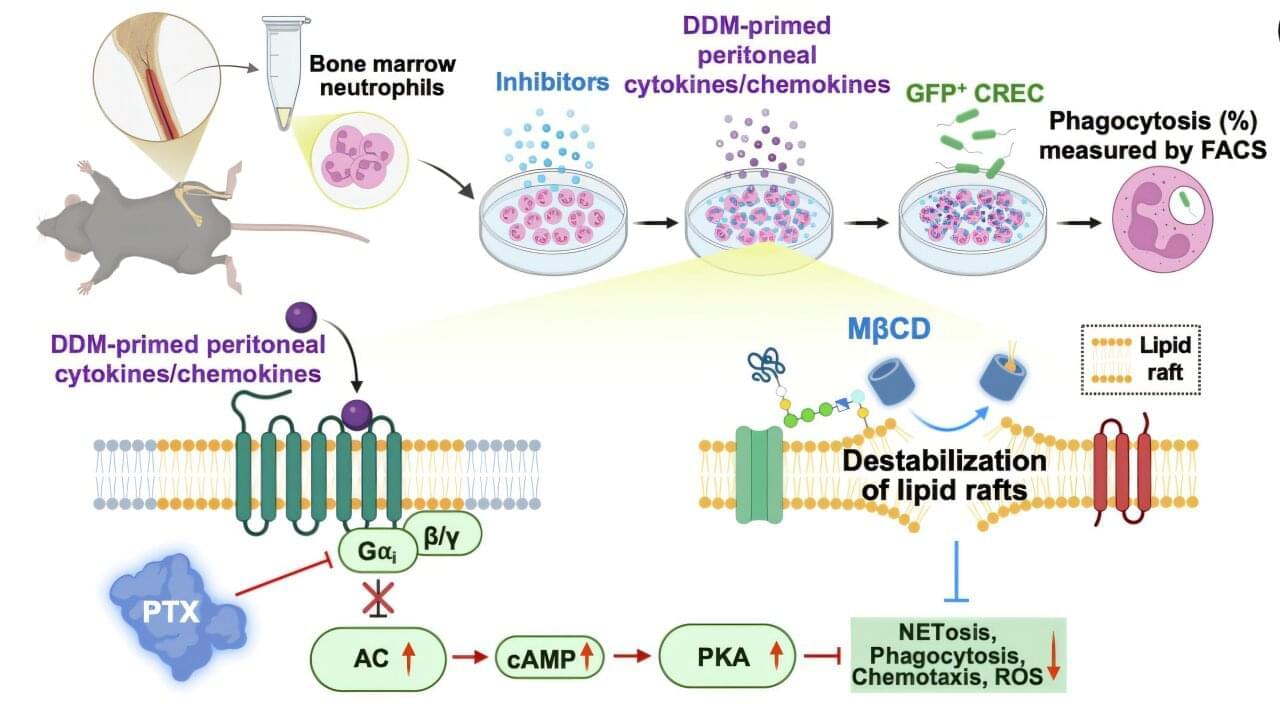
Secondary infections caused by bacteria or viruses during hospital care remain a long-standing global challenge, despite advances in modern medicine. In particular, mixed bacterial-viral infections in critically ill or immunocompromised patients are extremely difficult to treat and are associated with significantly increased mortality.
At the same time, the rapid rise of antibiotic-resistant bacteria and the frequent emergence of viral variants have exposed the limitations of existing antibiotics and vaccines. These challenges have driven growing interest in new strategies that prepare the body’s immune system in advance, enabling it to respond more rapidly and effectively when infection occurs.
Unlike conventional approaches that directly target specific pathogens, this emerging strategy focuses on priming the immune system so that immune cells can react faster and more strongly at the moment of infection.
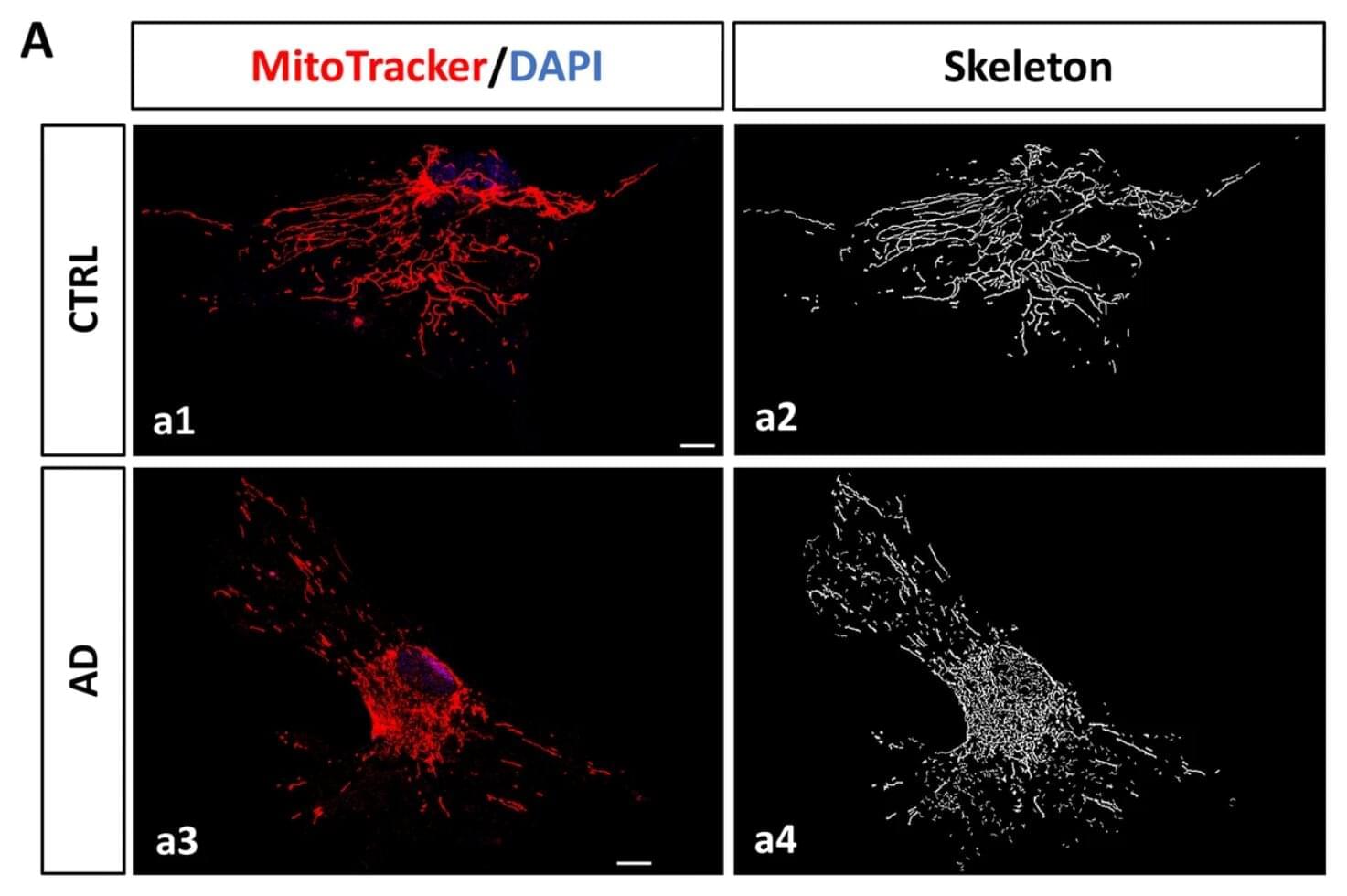
Researchers from the NeuroAD group (Neuropathology of Alzheimer’s Disease) within the Department of Cell Biology, Genetics and Physiology at the University of Málaga, also affiliated with IBIMA–BIONAND Platform and CIBERNED, have made a pioneering breakthrough in the fight against this disease by identifying astrocytes as a promising cellular target for the development of future therapies.
The study demonstrates, for the first time, the presence of senescent astrocytes—cells that remain alive but have lost their functional capacity—in the brains of Alzheimer’s patients, positioning this cellular aging process as a key mechanism in neurodegeneration.
The research, published in the journal Journal of Neuroinflammation, was led by Dr. Antonia Gutiérrez, Professor of Cell Biology and Principal Investigator of the NeuroAD group, together with Dr. Juan Antonio García León, Associate Professor of Cell Biology. Other contributors to the study include Laura Cáceres, Laura Trujillo, Elba López, Elisabeth Sánchez, and Inés Moreno.
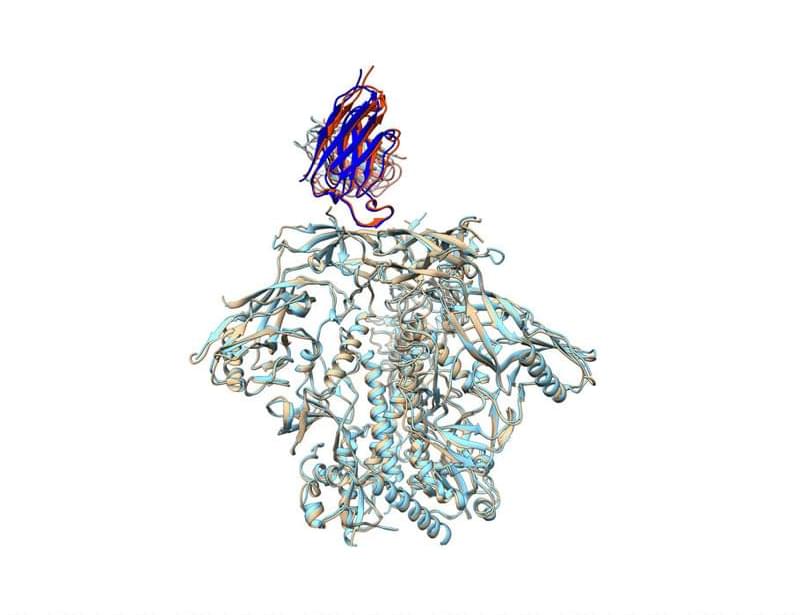
Two new animal studies show that B cell priming is key to induce broadly neutralizing antibodies (bNAbs) against HIV and demonstrate that a single immunization that targets bNAb precursors can induce potent neutralization. Learn more in Science Immunology:
B cell priming is a primary bottleneck to HIV-1 V2 apex bNAb elicitation.
Industrial yeasts are a powerhouse of protein production, used to manufacture vaccines, biopharmaceuticals, and other useful compounds. In a new study, MIT chemical engineers have harnessed artificial intelligence to optimize the development of new protein manufacturing processes, which could reduce the overall costs of developing and manufacturing these drugs.
Using a large language model (LLM), the MIT team analyzed the genetic code of the industrial yeast Komagataella phaffii — specifically, the codons that it uses. There are multiple possible codons, or three-letter DNA sequences, that can be used to encode a particular amino acid, and the patterns of codon usage are different for every organism.
The new MIT model learned those patterns for K. phaffii and then used them to predict which codons would work best for manufacturing a given protein. This allowed the researchers to boost the efficiency of the yeast’s production of six different proteins, including human growth hormone and a monoclonal antibody used to treat cancer.
A company called Fractal Health is developing a one-time GLP-1 gene therapy and the first humans are being dosed this year.
Instead of weekly injections like semaglutide or tirzepatide, this approach delivers genes directly into pancreatic beta cells. The therapy is controlled by the insulin promoter, meaning GLP-1 is only released when you eat — not continuously.
That could mean fewer systemic side effects and a more physiologic response.
They’re developing two versions:
• GLP-1 alone.
• GLP-1 + GIP (similar to tirzepatide)
One injection. Potentially permanent metabolic support.
If this works, it could redefine obesity and diabetes treatment.
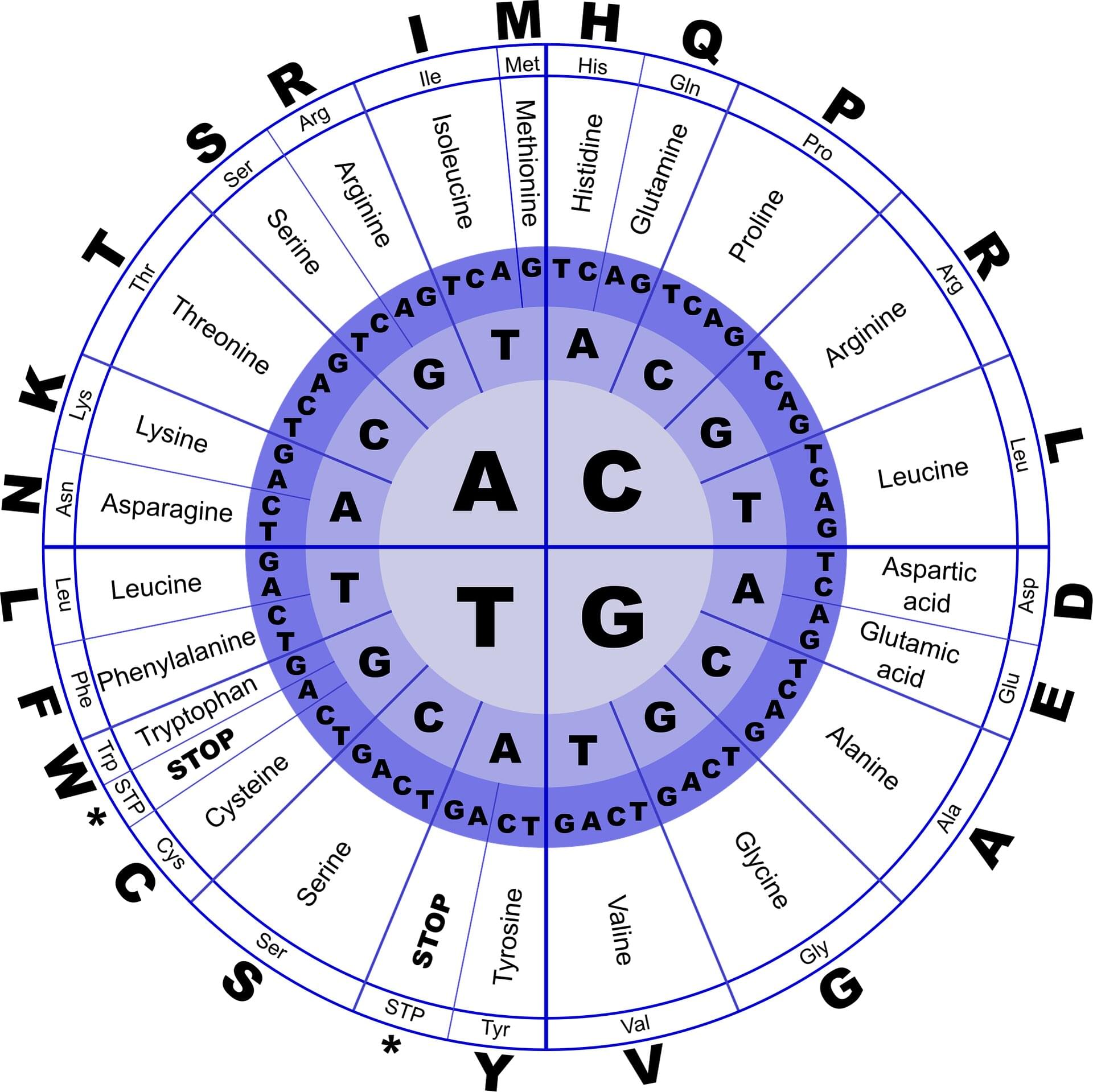
Industrial yeasts are a powerhouse of protein production, used to manufacture vaccines, biopharmaceuticals, and other useful compounds. In a new study, MIT chemical engineers have harnessed artificial intelligence to optimize the development of new protein manufacturing processes, which could reduce the overall costs of developing and manufacturing these drugs.
Using a large language model (LLM), the MIT team analyzed the genetic code of the industrial yeast Komagataella phaffii—specifically, the codons that it uses. There are multiple possible codons, or three-letter DNA sequences, that can be used to encode a particular amino acid, and the patterns of codon usage are different for every organism.
The new MIT model learned those patterns for K. phaffii and then used them to predict which codons would work best for manufacturing a given protein. This allowed the researchers to boost the efficiency of the yeast’s production of six different proteins, including human growth hormone and a monoclonal antibody used to treat cancer.