A new study develops ‘environment-modulating’ drugs that reinvigorate T-cells to once again destroy cancer cells.
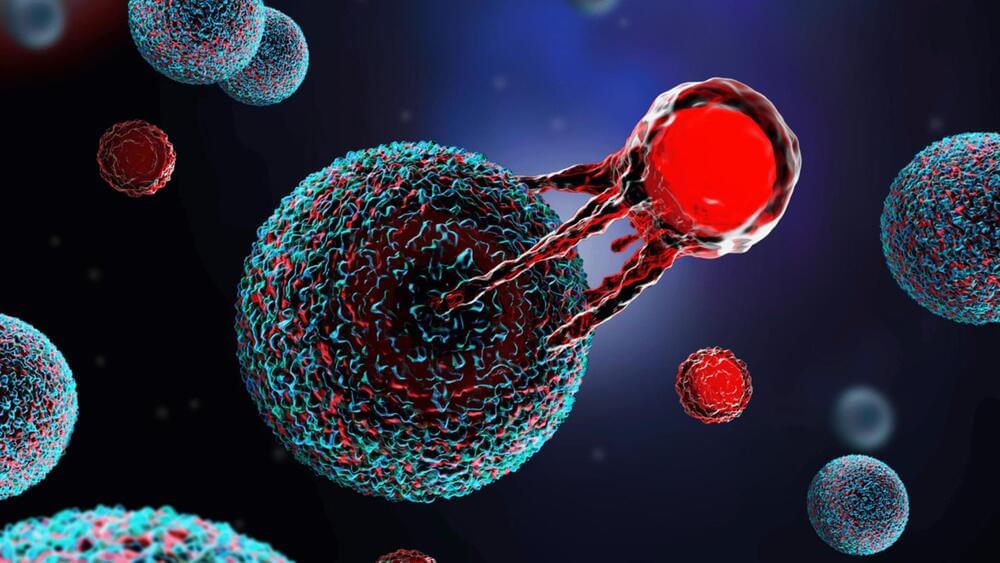
T-cells, typically thought to be anti-cancer, can switch sides and work against us in the right environment (or battlefield!), according to a new study published on December 21 in Nature.
This discovery is unexpected because many extensive studies before this believed that most worn-out T-cells’ properties were “irreversible.” Simply put, they were doomed to being subpar tumor killers.
Meletios Verras/iStock.
In light of this, the study went a step further by proving that altering the local environment around the tumor could “reinvigorate” T-cells to once again destroy cancer cells.