Researchers found that the body’s lymphatic drainage system may play an unexpected role in chronic organ rejection.


Individual variability in synaptic gene expression and synapse density in induced pluripotent stem cell–derived neurons predicted macro-scale alterations in gray matter volume and gamma-band activity in patients with Schizophrenia.
SIRS2026.
This genetic association study tests whether genetically driven variability in excitatory neurons’ transcriptome and synapse density in patient-derived neurons in vitro explain individual changes in cortical morphology, electrophysiology, and cognitive impairments in vivo.
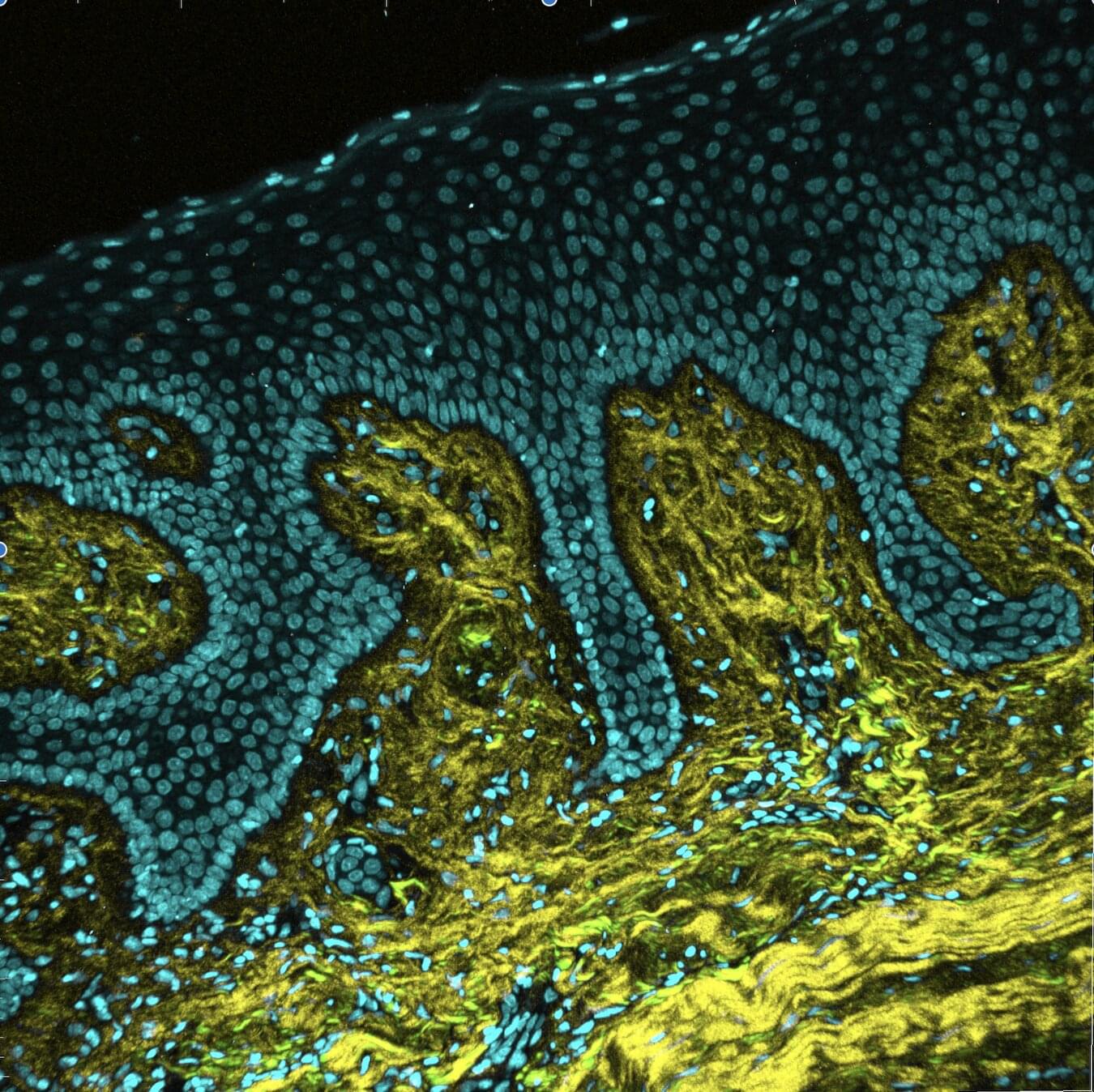
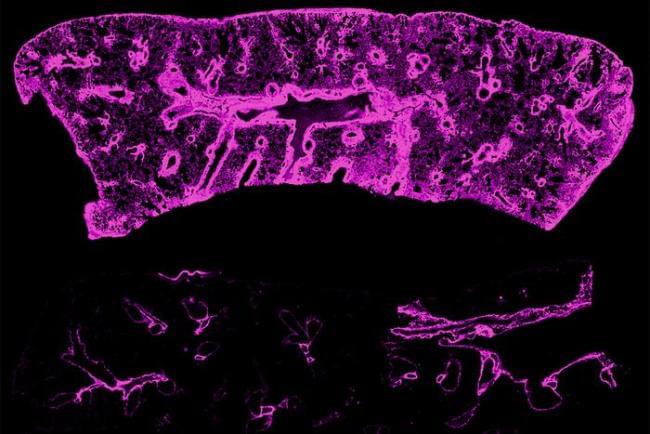
Periodontitis is a serious chronic inflammatory form of gum disease that affects millions worldwide. It can lead to tooth loss and the destruction of supporting bone. This disease has also been linked to other health problems, including diabetes, respiratory infections, and heart disease, impacting quality of life and increasing health care costs.
Current treatments target bacterial infection and inflammation through nonsurgical therapies, such as scaling and root planing, commonly known as “deep cleaning.” However, they do not repair the gum’s extracellular matrix (ECM), the gingival tissue’s structural support that is damaged by chronic inflammation. Without this foundation, gingival tissue cannot function properly, allowing inflammation to persist and slowing healing.
Now, new research led by Kyle H. Vining and Hardik Makkar of the School of Dental Medicine demonstrates how the physical properties of the gingival tissue impact periodontal health and disease. Their findings are published in Advanced Materials.
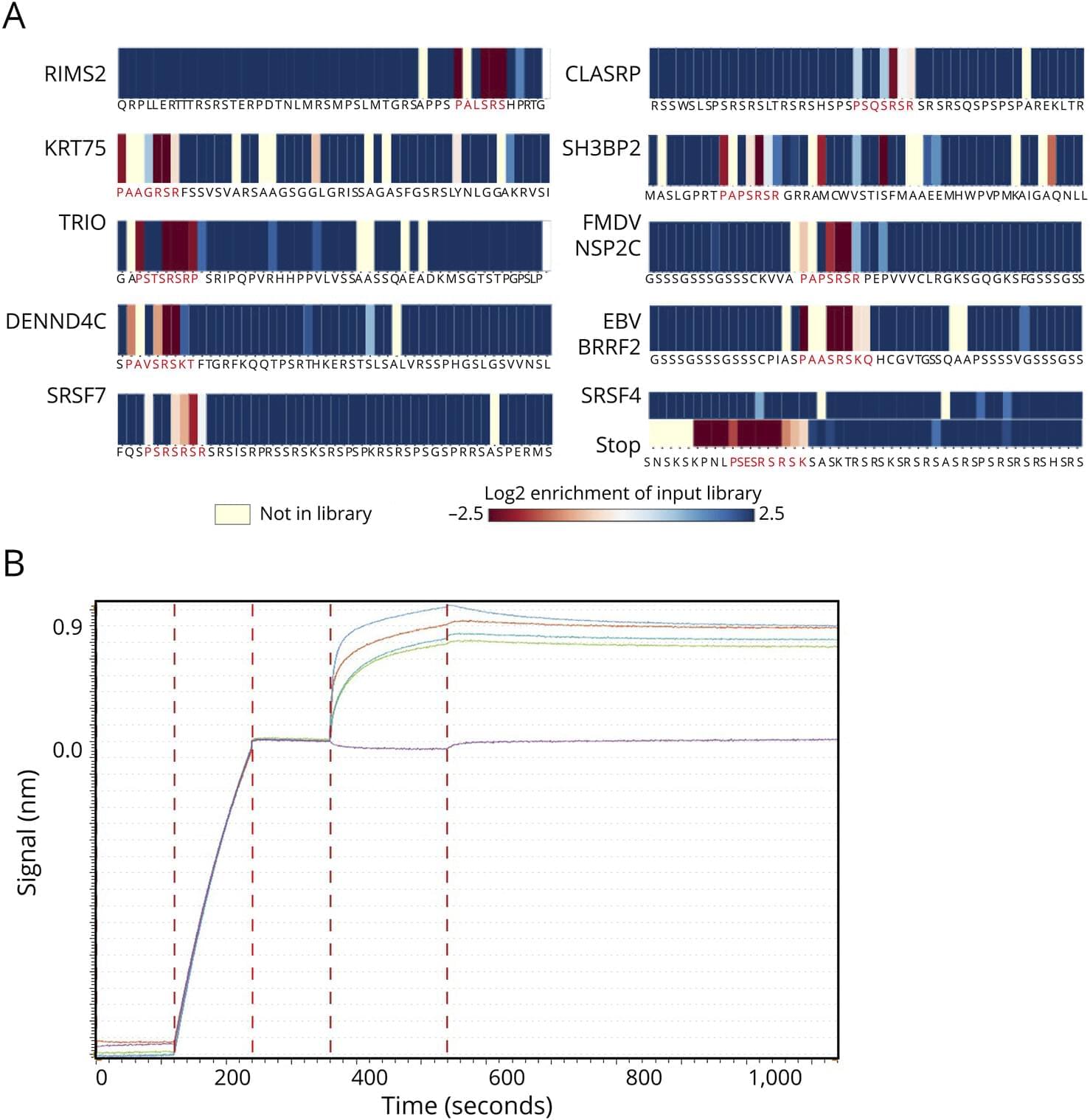

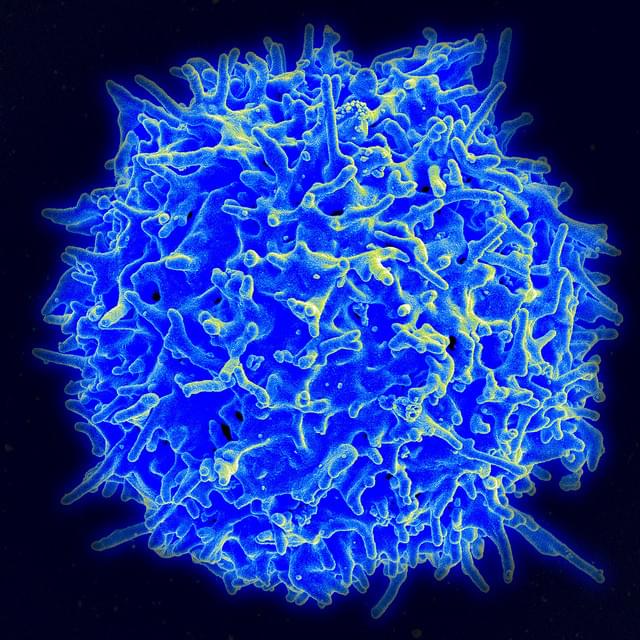
Signaling is fundamental to how cells sense and respond to their environment—but in immune cells, those signals must be precisely amplified to mount an effective defense against invasive threats. New research by immunologists in Germany is shedding light on how that amplification occurs in T cells, revealing a key molecular mechanism that helps trigger immune responses—and may also contribute to inflammatory conditions.
Writing in Science Signaling, researchers at the University Medical Center Hamburg-Eppendorf identified a crucial step in the production of a “second messenger,” an internal signal that relays and amplifies messages received at the cell surface. Because external signaling molecules cannot enter the cell, second messengers translate those cues into powerful intracellular responses.
In T cells, that process depends on NAADP (nicotinic acid adenine dinucleotide phosphate), a molecule that drives calcium (Ca²⁺) signaling—an essential step in T cell activation. Without it, T cells cannot become the effector cells needed to fight serious threats, such as infections or cancer.

Why are some people unable to hear from birth, even though their inner ear appears intact? One possible cause lies in the so-called OTOF gene. It plays a central role in transmitting sound signals from the hair cells to the auditory nerve. Without this function, acoustic information does not reach the brain.
Researchers from the German Primate Center—Leibniz Institute for Primate Research, the University Medical Center Göttingen, and the Max Planck Institute for Multidisciplinary Sciences have now, for the first time, generated marmosets in which this gene has been knocked out precisely. The animals are healthy and develop normally, but are deaf from birth. This provides the first primate model that realistically replicates key characteristics of human deafness. The results are published in Nature Communications.
Hearing loss is one of the most common congenital sensory disorders in humans. A major cause is a defect in the OTOF gene. This gene ensures that the protein otoferlin is produced in the inner ear. This protein is necessary for sound signals to travel from the hair cells to the auditory nerve. Without it, the ear still functions externally, but the signals do not reach the brain.
In this large, international cohort, contrast-associated acute kidney injury occurred in approximately 1 in 20 endovascular thrombectomy-treated patients with acute ischemic stroke and was independently associated with poor outcomes. A simple preprocedural risk score enables early identification of high-risk individuals and may support preventive strategies.
Background and Objectives.
POEMS (Polyneuropathy, Organomegaly, Endocrinopathy, M-protein and Skin changes) syndrome is a rare multisystem disorder where early identification is essential for better long term outcomes. Yet it is often misdiagnosed. Gonçalves et al review the condition here:
https://jnnp.bmj.com/content/early/2026/01/30/jnnp-2025-…e=facebook.
And this is a related editorial: https://jnnp.bmj.com/content/early/2026/01/30/jnnp-2025-…e=facebook
Polyneuropathy, Organomegaly, Endocrinopathy, M-protein and Skin changes (POEMS) syndrome is a rare multisystemic disorder associated with plasma cell dyscrasia, most commonly presenting with peripheral neuropathy. Due to its complex and heterogeneous clinical presentation, misdiagnosis is frequent, particularly with chronic inflammatory demyelinating polyradiculoneuropathy, which often leads to delays in appropriate management. Peripheral nerve involvement in POEMS syndrome is predominantly demyelinating, typically accompanied by early axonal degeneration. Specific clinical, neurophysiological and imaging features are key to differentiating POEMS from other acquired demyelinating neuropathies.
Drugs that block enzymes called tyrosine kinases are among the most effective targeted therapies for cancer. However, they typically work for only 40 to 80 percent of the patients who would be expected to respond to them.
In a new study, MIT researchers have figured out why those drugs don’t work in all cases: Many of these tumors have turned on a backup survival pathway that helps them keep growing when the targeted pathway is knocked out.
“This seems to be hardwired into the cells and seems to be providing activation of a critical survival pathway in cancer cells,” says Forest White, the Ned C. and Janet C. Rice Professor of Biological Engineering at MIT. “This pathway allows the cells to be resistant to a wide variety of therapies, including chemotherapies.”