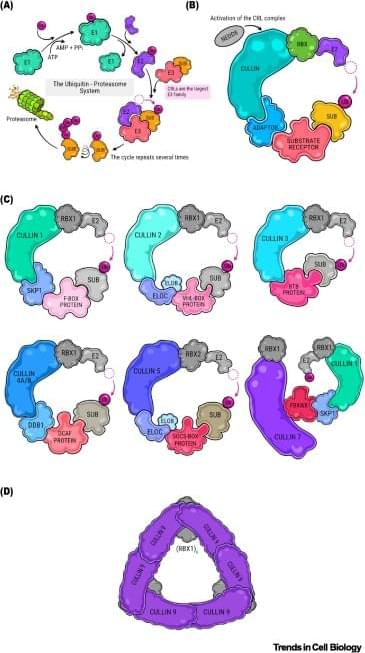
The researchers present the first integrative catalogue of 267 cullin–RING substrate receptors, of which 93 are linked to germline disorders.
The most frequent substrate receptor (SR)-related diseases are neurodevelopmental, neuromuscular, and congenital organ/skeletal syndromes.
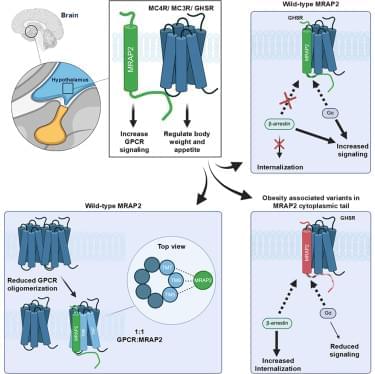
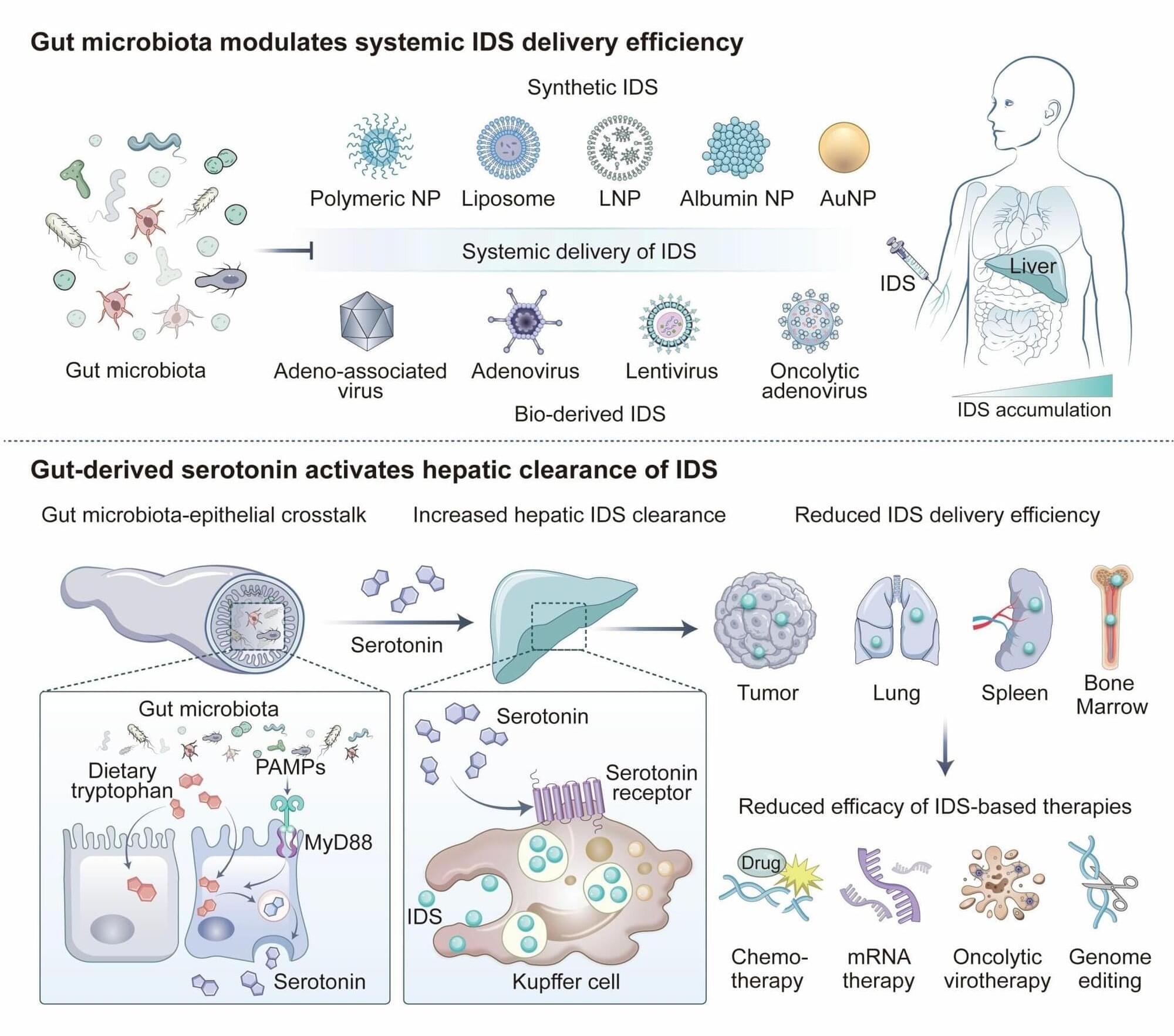
Disease associations are shaped by substrate context rather than tissue enriched expression.
Pathogenicity arises through altered degron recognition, disrupted complex assembly, dosage imbalance, or ubiquitin–proteasome system-independent functions.
Distinct variants in the same SR can yield divergent phenotypes, reflecting dosage sensitivity and developmental context.
Patient alleles inform diagnosis and therapeutic strategies, positioning SRs as central nodes connecting proteostasis, rare-disease genetics, and targeted protein degradation. sciencenewshighlights ScienceMission https://sciencemission.com/Cullin%E2%80%93RING-receptors