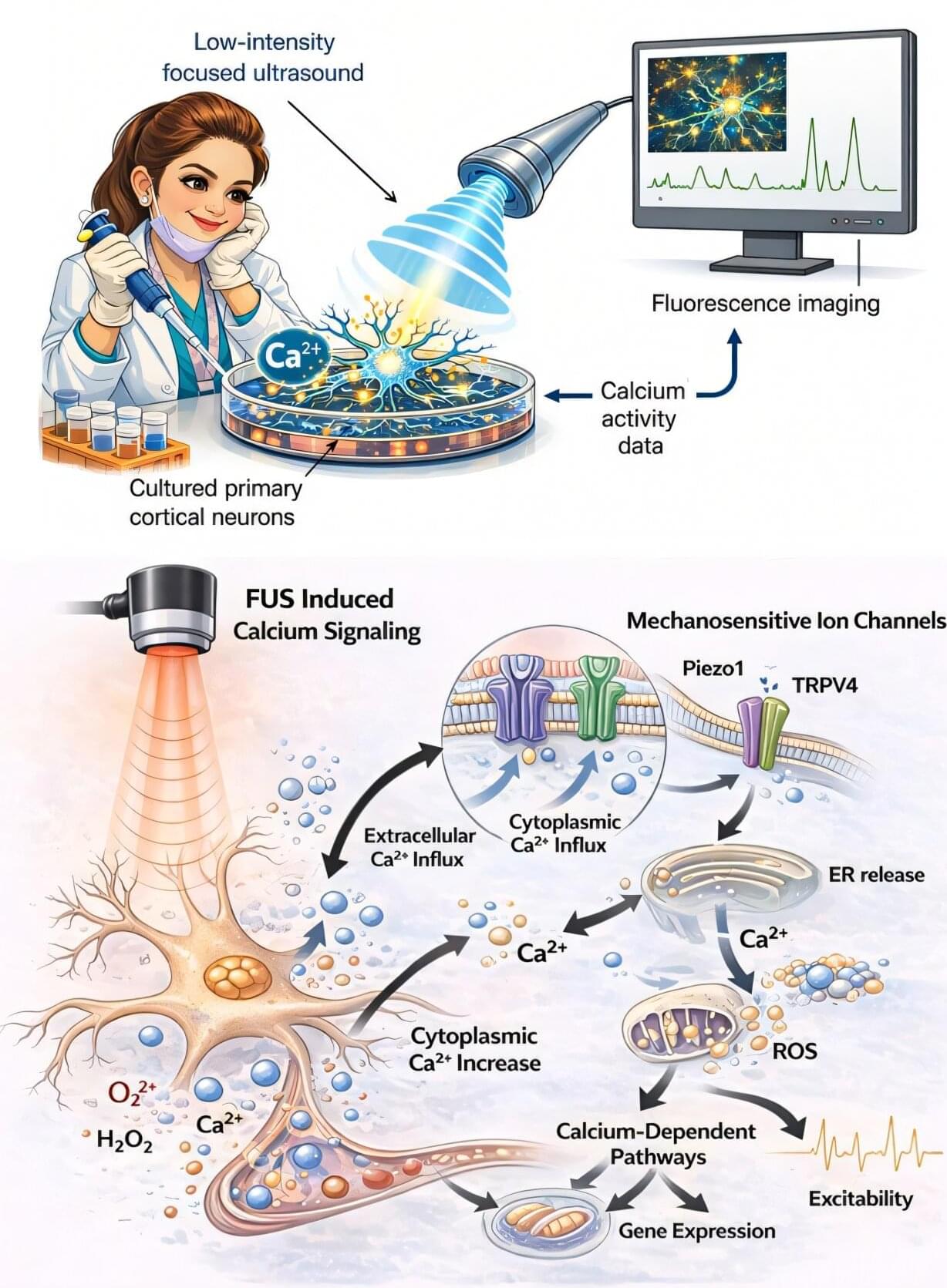
A pioneering study marks a major step toward eliminating the need for daily insulin injections for people with diabetes. The study was led by Assistant Professor Shady Farah of the Faculty of Chemical Engineering at the Technion—Israel Institute of Technology, in co-correspondence with MIT, and in collaboration with Harvard University, Johns Hopkins University, and the University of Massachusetts. The findings are published in the journal Science Translational Medicine.
The research introduces a living, cell-based implant that can function as an autonomous artificial pancreas, essentially a living drug that is long-term, thanks to a novel crystalline shield-protecting technology. Once implanted, the system operates entirely on its own: it continuously senses blood-glucose levels, produces insulin within the implant itself, and releases the exact amount needed—precisely when it is needed. In effect, the implant becomes a self-regulating, drug-manufacturing organ inside the body, requiring no external pumps, injections, or patient intervention.
One of the study’s most significant breakthroughs addresses the longstanding challenge of immune rejection, which has limited the success of cell-based therapies for decades. The researchers developed engineered therapeutic crystals—called “crystalline shield”—that shield the implant from the immune system, preventing it from being recognized as a foreign object. This protective strategy enables the implant to function reliably and continuously for several years.