This article reviews 12 interventions that the evidence suggests may help protect our brains as we age.


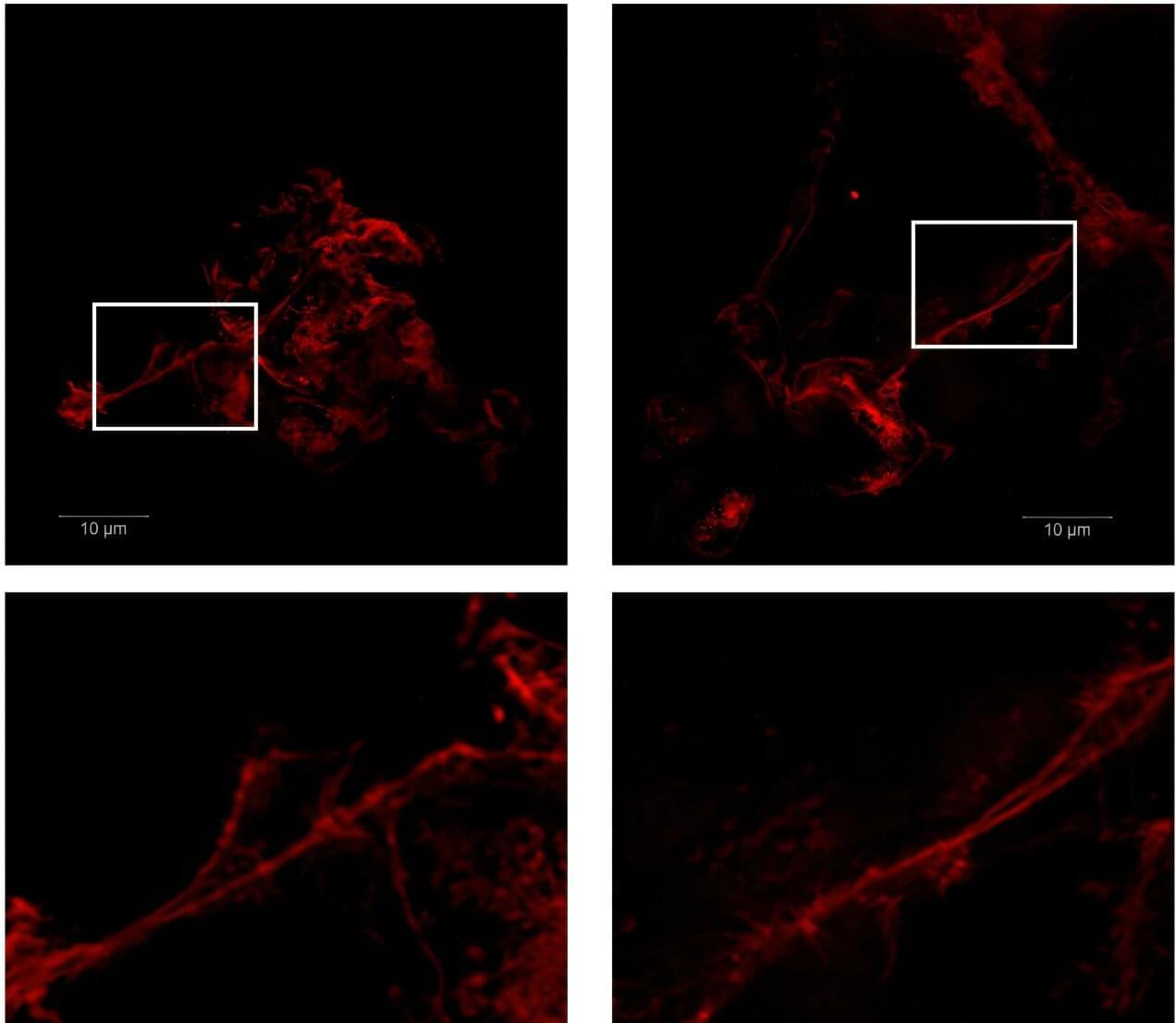
A new study led by researchers from VIB and KU Leuven shows that immune cells called microglia can actively promote the formation of plaques in Alzheimer’s disease, challenging the long-standing view that these cells serve only as defenders against plaque buildup. The findings were recently published in the Proceedings of the National Academy of Sciences.
“Most studies suggest that microglia are there to clean up the brain and remove the amyloid plaques. What we discovered is that actually they’re part of the problem. They generate plaques,” says Prof. Joost Schymkowitz, co-senior author of the study at the VIB-KU Leuven Center for Neuroscience. “It was thought that plaques aggregate by themselves. And it seems that the microglia, by trying to deal with the problem, amplify it.”
Alzheimer’s disease affects nearly 55 million people worldwide and is characterized by the accumulation of toxic protein aggregates in the brain known as amyloid plaques. These plaques are associated with neuronal death and progressive dementia. The brain’s microglia have been hailed as protectors against plaque accumulation in the disease, being the focus of several therapies. Nonetheless, the study shows how microglia are active producers of amyloid plaques in the earlier stages of the disease, reconsidering the therapeutic paradigm for Alzheimer’s.

Triggering receptor expressed on myeloid cells 2 (TREM2) is a cell surface transmembrane receptor from the TREM receptor family, predominantly expressed on the microglia in the central nervous system (CNS). TREM2-initiated signaling plays a crucial role in regulating neuroinflammation and neurodegeneration, particularly in the context of neurodegenerative diseases such as Alzheimer’s disease (AD) and Parkinson’s disease (PD), through the activation of downstream signaling pathways and transcriptional regulation of relevant genes. In this review, we aim to provide a concise review of the role and mechanistic implications of TREM2 in neurodegeneration and neuroinflammation, with a specific focus on AD and PD. We will discuss the most recent preclinical studies to highlight current advancements in the field. This review is intended to support both basic researchers and clinicians by enhancing their understanding of microglial function in the pathophysiology of AD and PD, as well as its role in neuroinflammation and neurodegeneration. Ultimately, we hope this contribution will pave the way for new discoveries and the development of potential therapeutic interventions.
© 2026. The Author(s).
Antibody titers elicited by the current inactivated influenza vaccine were modest against #Influenza A(H3N2) subclade K compared to responses against prior H1N1 and H3N2 strains.
Lower baseline and postvaccination titers to subclade K suggest partial immune evasion, raising implications for continued transmission and the selection of upcoming influenza vaccine strains.
This cohort study examines antibody responses to influenza A(H3N2) subclade K and other strains before and after vaccination against influenza.
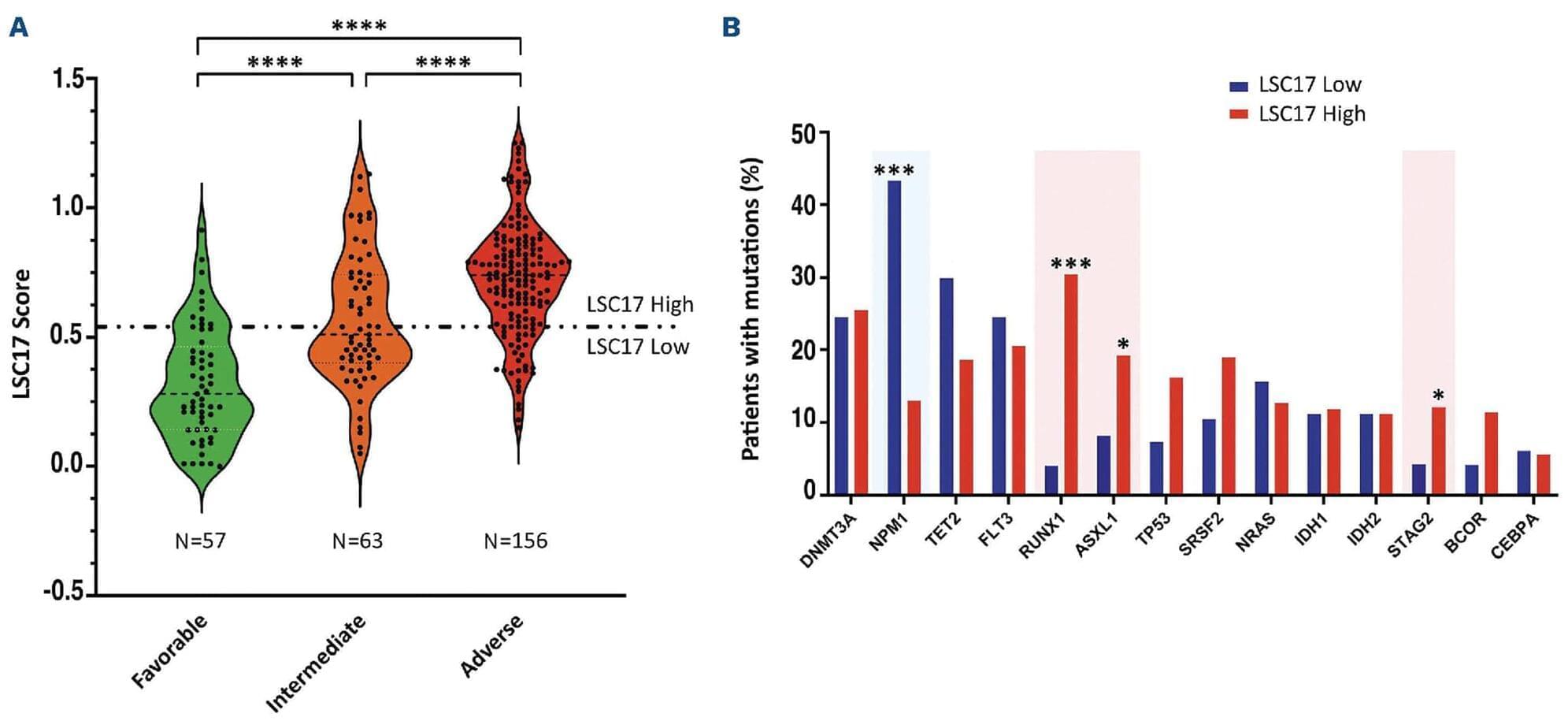
The identification of patients with acute myeloid leukemia (AML) who may have resistant disease when treated with standard induction chemotherapy is still challenging: Murphy and colleagues present the first prospective, multicenter study aiming to evaluate the prognostic value of the leukemic stem cell 17-gene (LSC17) score in patients with newly diagnosed AML.
Acute myeloid leukemia (AML) patients exhibit diverse molecular and cytogenetic changes with heterogeneous outcomes. The functionally-derived LSC17 gene expression score has demonstrated strong prognostic significance in retrospective analyses of adult and pediatric AML cohorts, where above-median scores are associated with worse outcomes compared to below-median scores in intensively-treated patients. Here we used a laboratory-developed clinically-validated NanoString-based LSC17 assay to test the prognostic value of the LSC17 score in a prospective multicenter study of 276 newly-diagnosed AML patients. Each patient’s score was classified as high or low by comparison to a previously-established reference score. In the entire cohort, a high LSC17 score was associated with poor risk features, including advanced age and unfavorable genetic mutations. In the subset of 190 patients treated intensively, a high LSC17 score was associated with lower remission rates (63% vs. 94% after induction; P0.0001), presence of measurable residual disease (46% vs. 10%; P0.0001), and shorter overall survival (OS, 606 days vs. not reached; P=0.0004; hazard ratio
Acute myeloid leukemia (AML) is a heterogeneous malignancy with multiple subtypes and variable clinical outcomes driven by disease characteristics as well as the clinical status of the patient.1 2,3 While genomic classification has further rationalized risk stratification in AML, many challenges remain.4 The accurate assessment of survival outcomes in AML subtypes driven by various combinations of driver mutations and cytogenetic abnormalities presents a challenge to the treating physician.5
AML is sustained by a rare subpopulation of leukemia stem cells (LSC) believed to drive therapy resistance and relapse.6,7 The LSC17 gene expression score was developed based on functionally-defined LSC populations across the spectrum of AML subtypes.8 In multiple independent retrospective cohorts, the LSC17 score has been found to robustly discriminate between patients with significantly different outcomes.9–12 Higher-than-median LSC17 scores were associated with poor treatment response and survival outcomes in both uni-and multi-variable survival analyses, independent of commonly used prognostic factors including cytogenetic and molecular risk groups.
Mayo Clinic researchers have identified a drug-and-supplement combination therapy that is capable of reducing the harmful effects of senescent cells – also known as “zombie cells” – in diabetic kidney disease.
In eBioMedicine, a publication of The Lancet, the team reported that the combination of the cancer drug dasatanib and a naturally occurring substance known as quercetin decreased inflammation and boosted protective factors in the kidney.
Diabetic kidney disease affects more than 12 million people in the U.S. and is the leading cause of kidney failure. While newer treatments can delay loss of kidney function, there is currently no cure.
Brain vasculature in ischemic stroke.
Ischemic stroke induces dynamic cellular structural changes in the neurovascular unit, leading to disrupted structural integrity of the blood–brain barrier, neuronal degeneration, and responsive angiogenesis coordinated by endothelial cells, reactive astrocytes, and pericytes.
After ischemic stroke, the neurovascular coupling function of the neurovascular unit is also disrupted, manifested by the metabolic dysregulation of glucose, lipid/fatty acid, and amino acids.
Neurovascular unit dynamic structural remodeling and metabolic dysfunction following ischemic stroke show cellular states and spatiotemporal heterogeneities, revealing new perspectives on ischemic stroke pathogenesis and future therapeutic strategies.
Multidimensional approach aiming to repair neurovascular unit structural disorganization and restore metabolic homeostasis with cellular and spatiotemporal precision is the optimal therapeutic strategy for ischemic stroke. sciencenewshighlights ScienceMission https://sciencemission.com/neurovascular-unit-in-ischemia
The neurovascular unit (NVU) is a multicellular system functioning to maintain healthy brain homeostasis and regulate the exchange of essential elements between the blood and the brain. Recent studies have shown that, in response to ischemic stroke (IS), the NVU undergoes dynamic structural remodeling and metabolic dysfunction, revealing new features of IS pathogenesis. Recent breakthroughs in single-cell multiomics provide emerging evidence regarding the spatiotemporal heterogeneity of NVU responses to IS. To date, clinical treatments for IS-induced brain injury remain very limited. These new studies have advanced our knowledge of the dynamic cellular and molecular changes of the NVU after IS, paving the way for new therapeutic strategies.

In the realm of medical advancements, a universal vaccine that can protect against any pathogen has long been a Holy Grail—and about as elusive as a mythological vessel. But Stanford Medicine researchers and collaborators have taken an astonishing step forward in that quest, surprising even themselves.
In a new study in mice, they have developed a universal vaccine formula that protects against a wide range of respiratory viruses, bacteria and even allergens. The vaccine is delivered intranasally—such as through a nasal spray—and provides broad protection in the lungs for several months.
In the study, published in Science, researchers show that vaccinated mice were protected against SARS-CoV-2 and other coronaviruses, Staphylococcus aureus and Acinetobacter baumannii (common hospital-acquired infections), and house dust mites (a common allergen).
New research points to the shingles vaccine meaningfully reducing the number of people who develop either dementia or mild cognitive decline later in life https://econ.st/4s73gdz.
Illustration: Cristina Spanò
The evidence is surprisingly strong.