PSA and PSMA kinetics after PSMA-guided prostate SBRT with focal boost. Can the marker and uptake kinetics inform us of the good, the bad and the ugly? Read about it in the RedJournal @vedangmurthy @drmaneesh_singh @docpriyamvada @RadOncTMC
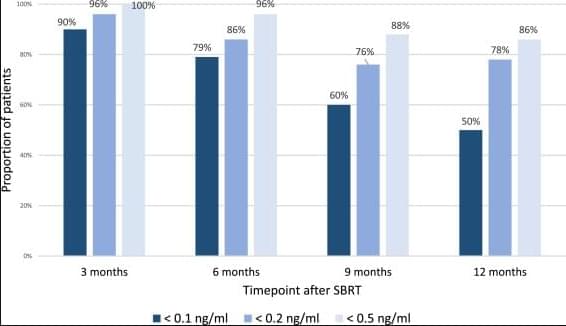
To evaluate PSA and PSMA kinetics following PSMA-PET and MR guided stereotactic body radiotherapy (SBRT) and short-term androgen deprivation therapy (ADT) with dominant intraprostatic lesion (DIL) boost in localised prostate cancer.