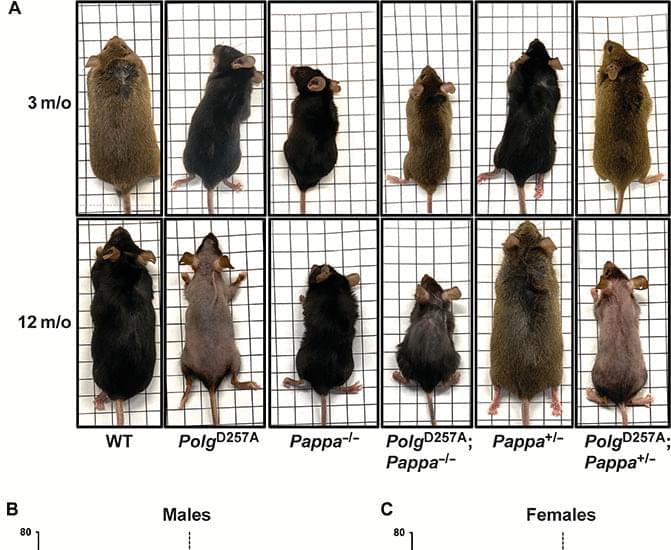
This insight has major implications for the development of antiaging therapies. First, they suggest that mtDNA integrity is not simply one of the many hallmarks of aging, but rather the foundation upon which others are built. And when that platform is broken, downstream hallmarks such as proteostasis or DNA repair cannot be engaged by typical means. Second, it suggests that interventions that target nutrient-sensing pathways may fail, or even backfire, when applied to organisms or tissues with high levels of mitochondrial damage. Hence, the next generation of geroprotective treatments must be tested in diverse models of aging, including those that combine multiple hallmarks, to better understand the scope and boundaries of their efficacy. Last, the efficacy of those treatments could be amplified by measures that improve the stability of the mitochondrial genome. While a reduction in IGF-1 signaling did not alter the frequency of mutations in WT or PolgD257A mice, it did slow the pace with which they reached homoplasmy. Thus, although it may not be possible today to reduce mitochondrial mutagenesis in human cells, our data show that it may already be possible to curtail the impact of mtDNA mutations on mammalian health span by slowing their clonal expansion in nondividing cells, the cells that are most sensitive to metabolic dysfunction.
While the precise mechanism by which Pappa influences clonal expansion of mtDNA mutations remains uncertain, several plausible explanations can be proposed. In the absence of cell division (the major driver for homoplasmy in dividing cells), the progression of mtDNA mutations toward homoplasmy is primarily driven by random genetic drift, the rate of mtDNA replication, and mitochondrial quality control. Thus, it is likely that loss of Pappa influences one of these three processes. Loss of Pappa may either reduce the rate of random genetic drift (potentially by changing mitochondrial fusion and fission or the spatial segregation of semi-isolated pockets of mtDNA), reduce the rate of mtDNA replication (less replication lowers the chance that a mutant mtDNA molecule expands enough to reach homoplasmy), or improve mitochondrial quality control by degrading mitochondria with mutant mtDNA molecules. It will be important to distinguish between these possibilities in future work to clear the way for novel interventions aimed at curbing the impact of mtDNA mutations on human health.
Regardless of the mechanism, these findings provide a compelling example of how the interplay between distinct hallmarks of the aging process can fundamentally alter the outcome of otherwise beneficial interventions. They reveal that the efficacy of antiaging strategies such as IGF-1 suppression is not absolute but context dependent. They are contingent on the integrity of underlying systems, including proteostasis and DNA repair. Without an intact mitochondrial genome, these pathways cannot be engaged, indicating that mtDNA integrity is required for these critical antiaging pathways. More broadly, our results underscore the need for a more integrated model of aging, one that considers not only individual pathways but also their interactions, hierarchies, and points of failure. By mapping these interactions, we can better anticipate the limitations of existing interventions and design next-generation therapies that are robust to the complex biology of aged tissues. In this light, strategies that target the expansion of mtDNA mutations, rather than their origin, may offer a powerful new axis for preserving tissue function and extending health span, even when the underlying genomic damage cannot be undone.