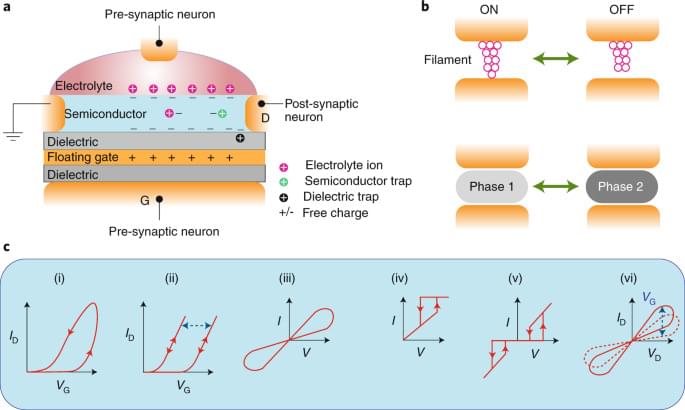
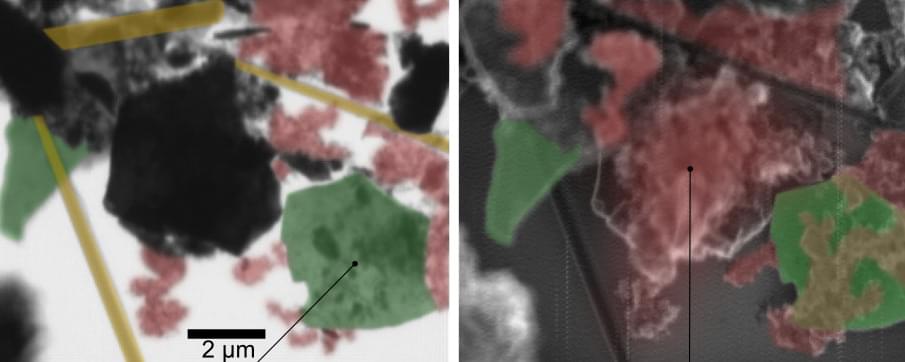
Memristive and nanoionic devices have recently emerged as leading candidates for neuromorphic computing architectures. While top-down fabrication based on conventional bulk materials has enabled many early neuromorphic devices and circuits, bottom-up approaches based on low-dimensional nanomaterials have shown novel device functionality that often better mimics a biological neuron. In addition, the chemical, structural and compositional tunability of low-dimensional nanomaterials coupled with the permutational flexibility enabled by van der Waals heterostructures offers significant opportunities for artificial neural networks. In this Review, we present a critical survey of emerging neuromorphic devices and architectures enabled by quantum dots, metal nanoparticles, polymers, nanotubes, nanowires, two-dimensional layered materials and van der Waals heterojunctions with a particular emphasis on bio-inspired device responses that are uniquely enabled by low-dimensional topology, quantum confinement and interfaces. We also provide a forward-looking perspective on the opportunities and challenges of neuromorphic nanoelectronic materials in comparison with more mature technologies based on traditional bulk electronic materials.