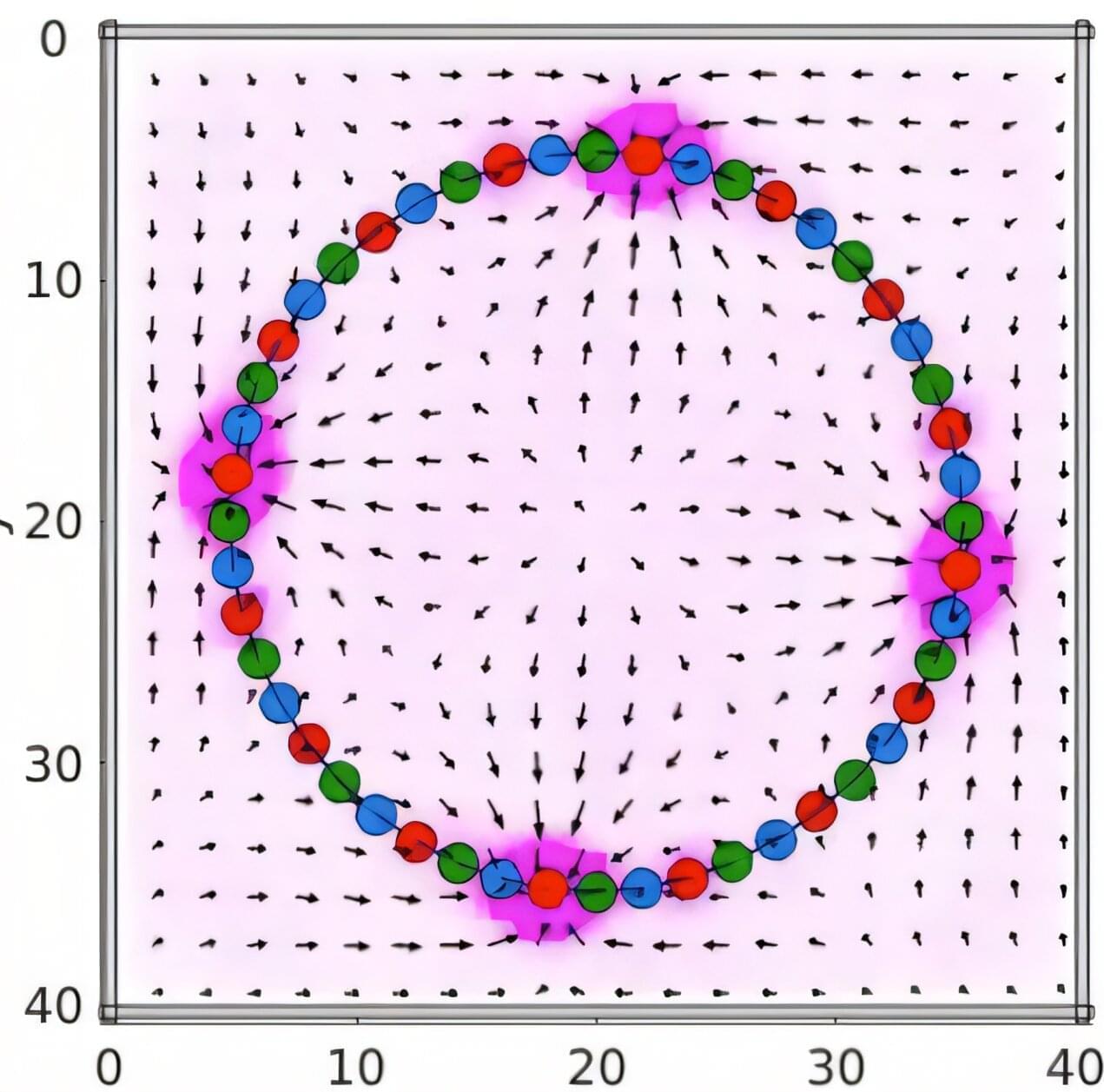
What if a soft material could move on its own, guided not by electronics or motors, but by the kind of rudimentary chemical signaling that powers the simplest organisms? Researchers at the University of Pittsburgh Swanson School of Engineering have modeled just that—a synthetic system that on its own directly transforms chemical reactions into mechanical motion, without the need for the complex biochemical machinery present in our bodies.
Just like jellyfish, some of the simplest organisms do not have a centralized brain or nervous system. Instead, they have a “nerve net” which consists of dispersed nerve cells that are interconnected by active junctions, which emit and receive chemical signals. Even without a central “processor,” the chemical signals spontaneously travel through the net and trigger the autonomous motion needed for organisms’ survival.
In a study published in PNAS Nexus, Oleg E. Shklyaev, research assistant, and Anna C. Balazs, Distinguished Professor of Chemical and Petroleum Engineering and the John A. Swanson Chair of Engineering, have developed computer simulations to design a soft material with a “nerve net” that links chemical and mechanical networks in a way that mimics how the earliest and simplest living systems coordinate motion.