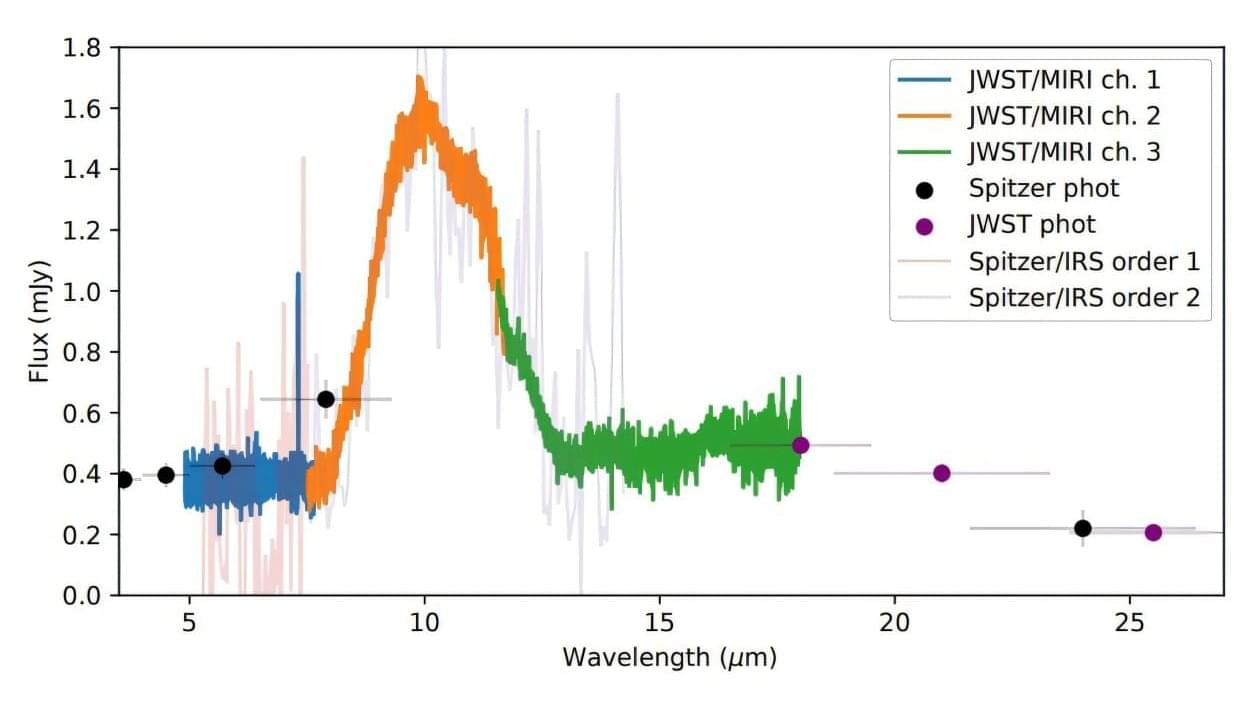
Using the James Webb Space Telescope (JWST), astronomers have performed infrared observations of a planetary debris disk around a nearby white dwarf known as GD 362. Results of the new observations, presented October 8 on the arXiv preprint server, yield important insights into the chemical composition of this disk.
White dwarfs (WDs) are stellar cores left behind after a star has exhausted its nuclear fuel. Due to their high gravity, they are known to have atmospheres of either pure hydrogen or pure helium.
However, there exists a small fraction of WDs that shows traces of heavier elements, and they are believed to be accreting planetary material. Studies of this material around WDs, which often forms dust disks, is essential to improving our knowledge of how planets form and evolve.