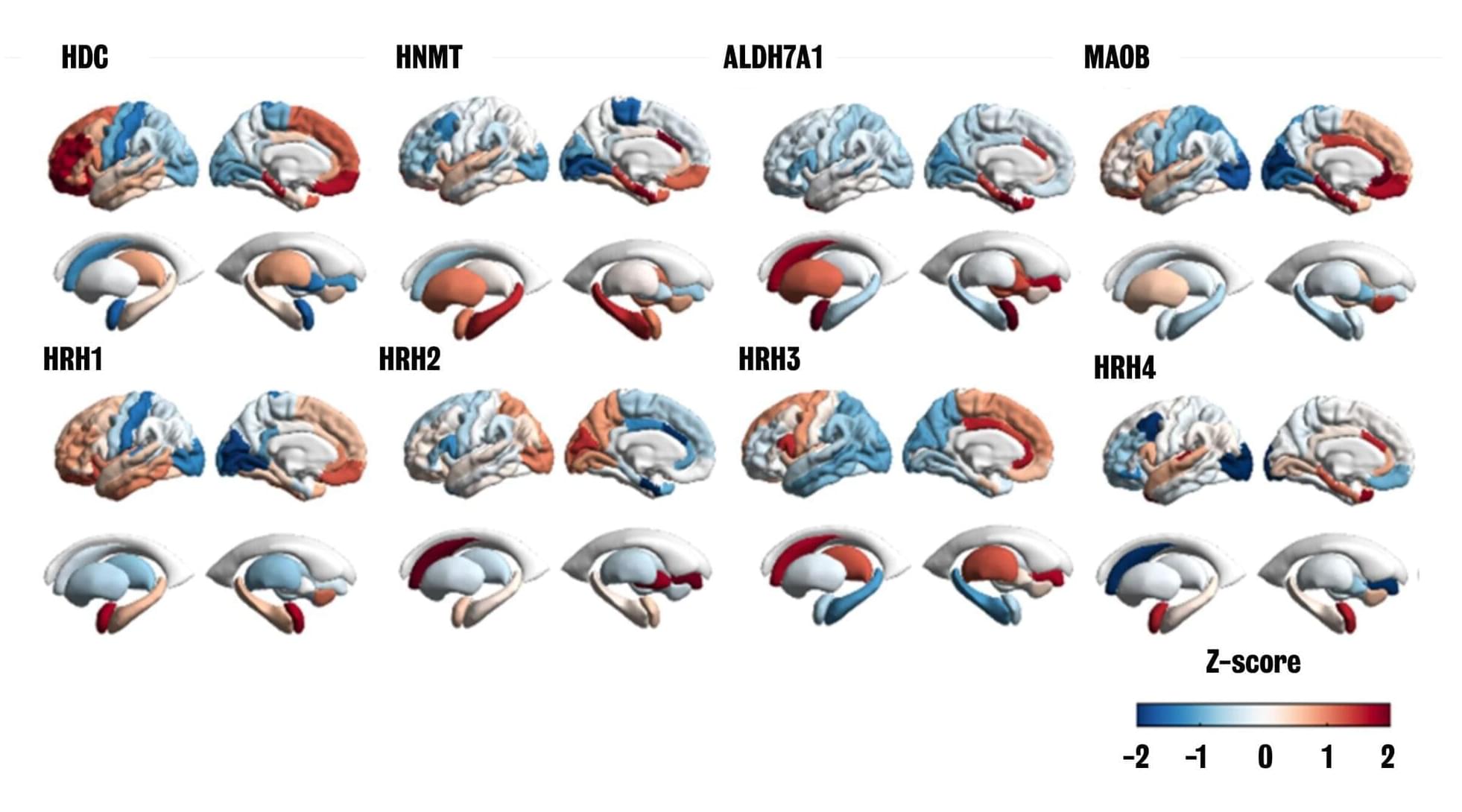
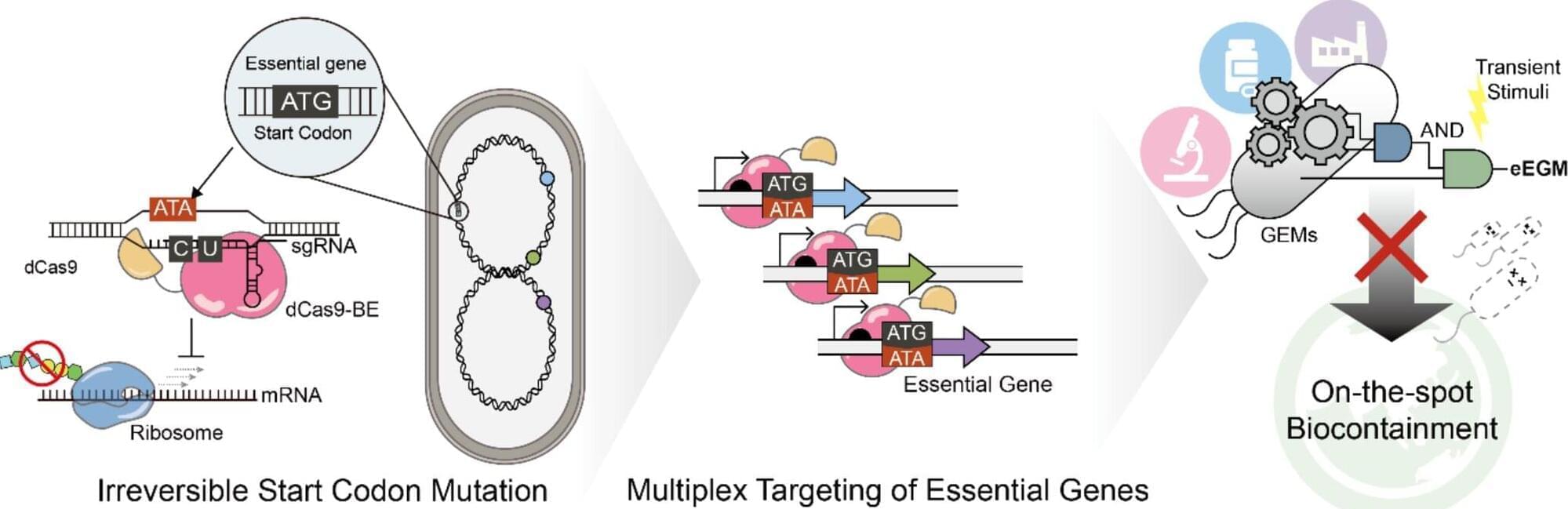
New research from King’s College London and the University of Porto has mapped the histamine system in the brain. Histamine, a molecule more commonly associated with allergies, plays a separate but poorly understood role in brain function. This study addresses this gap, building the first multiscale map of the histamine system that spans from genetics to behavior and related mental health conditions.
The findings provide a new framework for understanding how this often-overlooked chemical system contributes to brain function and could point toward new treatment strategies for histamine-related conditions such as depression, ADHD, and schizophrenia. The study is published in Nature Mental Health.
Histamine is a neurotransmitter, a molecule crucial for neurons to communicate with one another. Neuroscience research has classically focused on understanding other neurotransmitter systems such as dopamine and serotonin.