Olaf SpornsMay 1, 2015Annual Science Board Symposium — New Science. New Horizons.
Category: neuroscience – Page 11
Tech Meets Neuroscience with Adam Gazzaley
Dr. Adam Gazzaley, Cognitive Neuroscientist at the University of California San Francisco, explores the latest research on the interaction between the brain and technology.
The Thousand Brains Theory of Intelligence | Jeff Hawkins | Numenta
This presentation is clipped from a virtual keynote Jeff was invited to give on December 16.Numenta technology is built on the Thousand Brains Theory, our se…
Beyond Paradox | Iain McGilchrist
“There is nothing either good or bad, but thinking makes it so.”
— Hamlet, Act II, Scene 2
The two hemispheres of our brain collaborate to produce a coherent understanding of the world—at least, that’s what they’re supposed to do. In his groundbreaking book, The Master and His Emissary, neuro-philosopher and psychiatrist, Iain McGilchrist, proposed that our culture has been captured by the left hemisphere, whose dogmatic, technical and irrational way of processing information leads it to manifestly dangerous conclusions about the way the world works. Importantly, the left hemisphere never changes its mind.
In one of the widest conversations on Planet: Critical to date, Iain explains how we came to lose sight of the bigger picture by forsaking the intuition, creativity and intelligence of the right hemisphere. We discuss how our relationship to language makes and unmakes the world, the search for meaning, human agency, relationality, morality, art and the divine, with Iain clearly spelling out a path to human fulfilment—which may very well be the only thing which can save Earth from the worst of us.
🔴 The Master and His Emissary: https://channelmcgilchrist.com/master… Platformed: Charles Foster 🌎 Support Planet: Critical: / planetcritical 🌎 Subscribe: https://planetcritical.com/ 🌎 BlueSky: https://bsky.app/profile/racheldonald…
🗣️ Platformed: Charles Foster
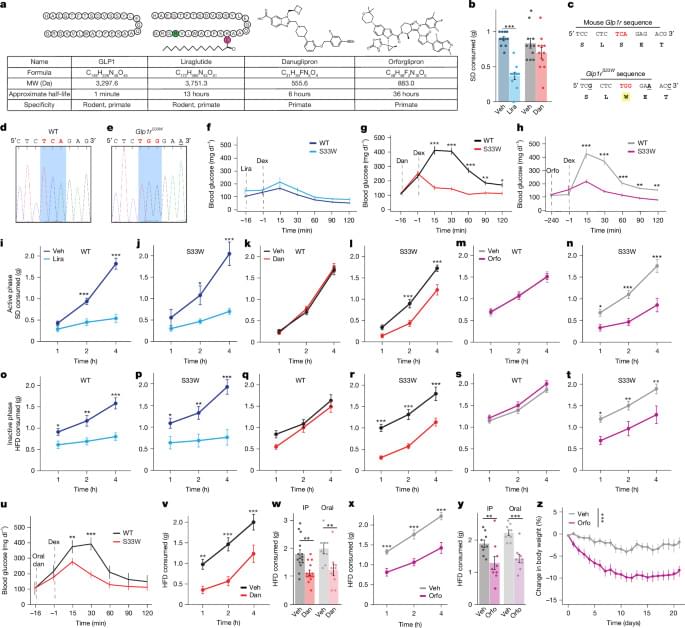
A brain reward circuit inhibited by next-generation weight-loss drugs in mice
New research using humanized mouse models has finally pulled back the curtain on how these small-molecule drugs work in the brain, revealing that they don’t just tell the body it’s full—they actually change how we perceive “treats.”
1. The Homeostatic Circuit: This is the body’s fuel gauge. It involves the hypothalamus and hindbrain, which manage basic hunger and energy levels. It’s the circuit that tells you, “I’ve had enough calories for today.”
These new weight-loss pills do more than just settle your stomach; they speak directly to the brain’s reward center to help quiet the “food noise” that leads to overeating.
This is a “proof of concept” study. It proves the mechanism exists, but it doesn’t yet guarantee that a pill will be a side-effect-free “cure” for overeating in humans.
Humanized glucagon-like peptide 1 receptor (GLP1R) mouse models are used to investigate the neural circuitry through which small-molecule GLP1R agonists modulate feeding, with implications for how these orally delivered weight-loss drugs engage brain reward circuits.
Nanotechnology-Driven Therapeutic Innovations in Neurodegenerative Disorders: A Focus on Alzheimer’s and Parkinson’s Disease
Neurodegenerative disorders entail a progressive loss of neurons in cerebral and peripheral tissues, coupled with the aggregation of proteins exhibiting altered physicochemical properties. Crucial to these conditions is the gradual degradation of the central nervous system, manifesting as impairments in mobility, aberrant behaviors, and cognitive deficits. Mechanisms such as proteotoxic stress, neuroinflammation, oxidative stress, and programmed cell death contribute to the ongoing dysfunction and demise of neurons. Presently, neurodegenerative diseases lack definitive cures, and available therapies primarily offer palliative relief. The integration of nanotechnology into medical practices has significantly augmented both treatment efficacy and diagnostic capabilities.

Researchers discover advanced language processing in the unconscious human brain
Baylor College of Medicine researchers have found that the human brain is capable of sophisticated language processing while in an unconscious state from general anesthesia. The findings, published in the latest edition of Nature, challenge what we know about the role of consciousness and cognition, and could open new ways of understanding memory, language and brain-computer interfaces.
“Our findings show that the brain is far more active and capable during unconsciousness than previously thought,” said Dr. Sameer Sheth, professor and Cullen Foundation Endowed chair of neurosurgery and a McNair Scholar at Baylor. “Even when patients are fully anesthetized, their brains continue to analyze the world around them.”
Sheth, who is also a neurosurgeon at Baylor St. Luke’s Medical Center, and his collaborators first recorded neural activity from hundreds of individual neurons in the hippocampus, a part of the brain associated with memory, while patients were under general anesthesia during epilepsy surgery. Patients undergoing this type of surgery were sought after because it allowed researchers access to this particular part of the brain.


ALS and Huntington Disease: Unraveling the Connections between TDP-43 and Huntingtin
Amyotrophic lateral sclerosis (ALS) and Huntington disease (HD) are lethal neurodegenerative diseases affecting motor function. Though their etiology and pathology are distinct, recent evidence suggests commonalities between TAR DNA-binding protein (TDP-43), which is associated with 97% of ALS cases, and huntingtin (HTT), the causative protein of HD. ALS is a heterogeneous, lethal neurodegenerative disease characterized by the progressive loss of upper and lower motor neurons, as well as brainstem and spinal cord degeneration. The causes of ALS are complex, variable, and, in some cases, unknown, but most cases involve mislocalization of the protein TDP-43. In contrast, HD is a monogenic, autosomal dominant, lethal neurodegenerative disease caused by polyglutamine expansion in HTT protein and characterized by the progressive loss of neurons in the brain, particularly in the striatum, which results in motor, cognitive, and behavioral changes. Although HD is not typically associated with motor neuron loss, recent evidence suggests a link between HTT and TDP-43 within the context of both ALS and HD, as well as links to related neurodegenerative diseases, such as frontotemporal dementia (FTD) and spinocerebellar ataxia type 2 (SCA2). Herein, we discuss confirmed cases of concurrent ALS and HD and the overlap of underlying disease mechanisms that potentially contribute to the onset and progression of these two devastating neurodegenerative diseases, with a focus on commonalities between TDP-43 and HTT. We propose that elucidating these commonalities will aid in the identification of broad-spectrum disease risk factors and potential overlapping treatment targets.
Temporal superposition and feature geometry of RNNs under memory demands
Abstract: Understanding how populations of neurons represent information is a central challenge across machine learning and neuroscience. Recent work in both fields has begun to characterize the representational geometry and functionality underlying complex distributed activity. For example, artificial neural networks trained on data with more features than neurons compress data by representing features non-orthogonally in so-called *superposition*. However, the effect of time (or memory), an additional capacity-constraining pressure, on underlying representational geometry in recurrent models is not well understood. Here, we study how memory demands affect representational geometry in recurrent neural networks (RNNs), introducing the concept of temporal superposition. We develop a theoretical framework in RNNs with linear recurrence trained on a delayed serial recall task to better understand how properties of the data, task demands, and network dimensionality lead to different representational strategies, and show that these insights generalize to nonlinear RNNs. Through this, we identify an effectively linear, dense regime and a sparse regime where RNNs utilize an interference-free space, characterized by a phase transition in the angular distribution of features and decrease in spectral radius. Finally, we analyze the interaction of spatial and temporal superposition to observe how RNNs mediate different representational tradeoffs. Overall, our work offers a mechanistic, geometric explanation of representational strategies RNNs learn, how they depend on capacity and task demands, and why.
Supplementary Material: zip
Primary Area: interpretability and explainable AI.