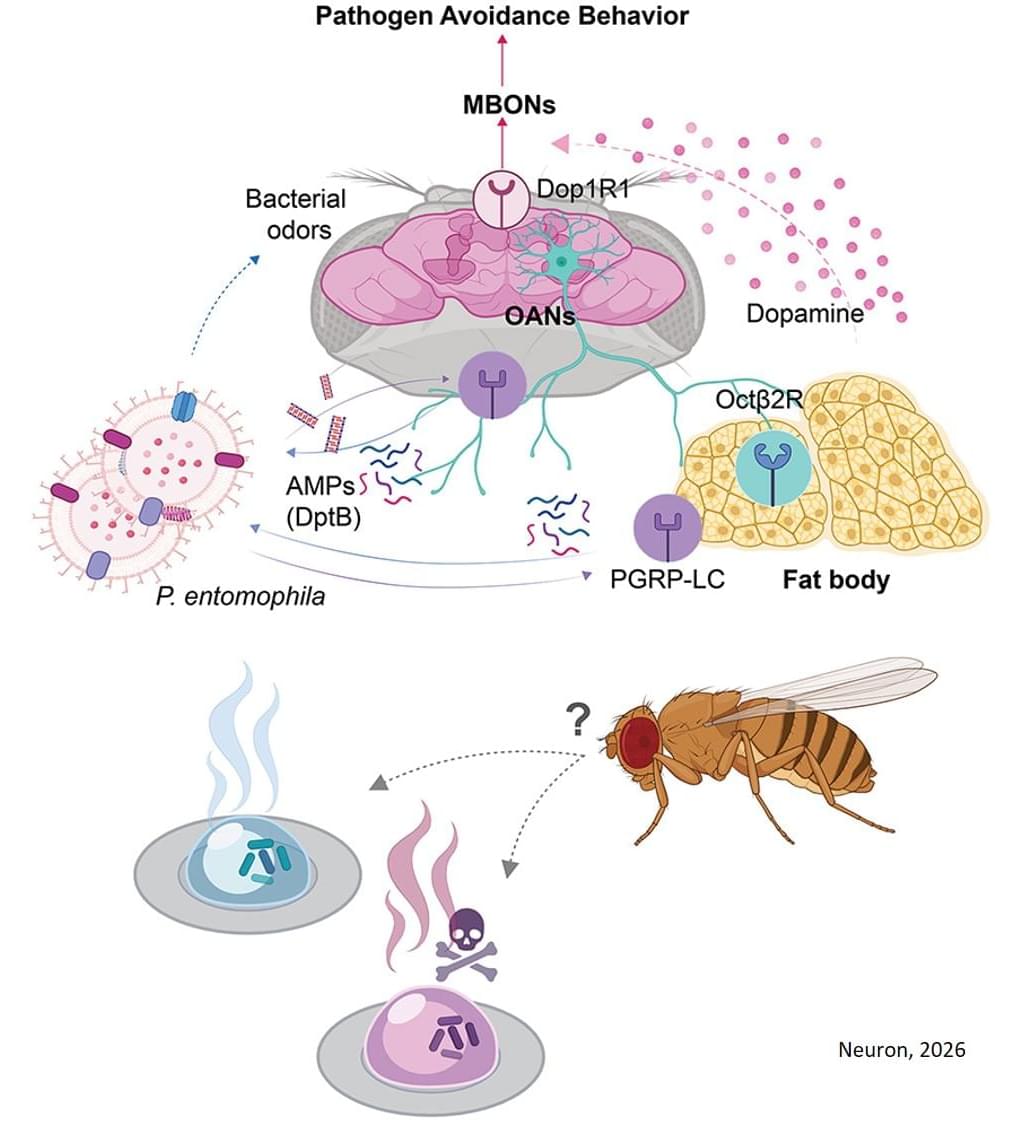
It is not yet known how the immune system’s discovery of the pathogens leads to a change in behavior. “As this learned food avoidance can be found in all species, we investigated this question in a model organism – the fruit fly Drosophila,” explains the senior author. “Within this model, we can clarify how the brain and body interact with each other to trigger an avoidance reaction that is vital for survival.”
In the current study, the group had their test animals choose between two food sources. One of them was contaminated with the pathogenic bacterium Pseudomonas entomophila. The other contained a harmless Pseudomonas strain. The two food sources were otherwise completely identical.
Flies that have not yet had any bad experiences with the pathogen prefer the harmful food because they find its odor attractive. “As this is life-threatening for the animals, we wondered how animals that have consumed these bacteria with their food behave,” explains the scientist.
The pathogens did not remain undiscovered among the flies for long: The animals’ innate immune system has sensors that raise the alarm in cases such as this. “In our experiment, receptors were activated in them that respond to components of the bacterial cell wall,” explains another author.
These sensors mainly respond to the harmful Pseudomonas strain, but hardly respond at all to the harmless strain. Many of them sit on the surface of special neurons located near the fly’s throat. Via their branches, these neurons are connected not only to the fly’s brain but also to a fat store in the fly’s head. If the receptors raise the alarm in the presence of harmful microorganisms, this leads to the release of the neurotransmitter octopamine in the neurons, which is closely related to adrenaline. This travels through the neuronal branches to the fat store.
“The octopamine then triggers the formation of another neurotransmitter, dopamine, in the fat cells,” says the author. “The dopamine, in turn, is transported into the fly’s brain, where it causes the continuous, increased activation of neuronal networks that are important for learning and trigger an avoidance response.” The animals then tend to be deterred by the odor of pathogenic bacteria. “We were able to show that the flies chose the food source with the harmless germs following their experience with the spoiled food,” explains the scientist.
The adipose tissue is significantly involved in this learned behavioral change. But why is that so? “We still do not have a definitive answer,” says the author. “However, the flies’ decision may be linked to their nutritional status.”