Researchers have developed a groundbreaking 3D brain model that closely mirrors the architecture and function of the human brain.


Please consider joining my Substack at https://rupertsheldrake.substack.com.
Does Nature Obey Laws? | Sheldrake-Vernon Dialogue 95.
The conviction that the natural world is obedient, adhering to laws, is a widespread assumption of modern science. But where did this idea originate and what beliefs does it imply? In this episode of the Sheldrake-Vernon Dialogues, Rupert Sheldrake and Mark Vernon discuss the impact on science of the Elizabethan lawyer, Francis Bacon. His New Instrument of Thought, or Novum Organum, put laws at the centre of science and was intended as an upgrade on assumptions developed by Aristotle. But does the existence of mind-like laws of nature, somehow acting on otherwise mindless matter, even make sense? What difference is made by insights subsequent to Baconian philosophy, such as the discovery of evolution or the sense that the natural world is not machine-like but behaves like an organism? Could the laws of nature be more like habits? And what about the existence of miracles, the purposes of organisms, and the extraordinary fecundity of creativity?
—
Dr Rupert Sheldrake, PhD, is a biologist and author best known for his hypothesis of morphic resonance. At Cambridge University, as a Fellow of Clare College, he was Director of Studies in biochemistry and cell biology. As the Rosenheim Research Fellow of the Royal Society, he carried out research on the development of plants and the ageing of cells, and together with Philip Rubery discovered the mechanism of polar auxin transport. In India, he was Principal Plant Physiologist at the International Crops Research Institute for the Semi-Arid Tropics, where he helped develop new cropping systems now widely used by farmers. He is the author of more than 100 papers in peer-reviewed journals and his research contributions have been widely recognized by the academic community, earning him a notable h-index for numerous citations. On ResearchGate his Research Interest Score puts him among the top 4% of scientists.
https://www.sheldrake.org/about-rupert-sheldrake?svd=95
—

Children on the autism spectrum, often at odds with their own emotions, may find help in an unlikely place: their own inner voice. A team from the University of Pittsburgh and collaborators tested a novel intervention designed to train autistic children in developing internal speech—and the results suggest it may reduce emotional dysregulation.
Emotional outbursts and difficulty with self-control can disrupt the lives of many autistic children and their families. Traditional therapies rarely focus on the language children use with themselves, the internal dialogue that helps regulate emotion and behavior in neurotypical development.
Prior research has shown inner speech supports problem-solving and self-regulation, yet many autistic individuals seem to lack this internal toolset.

The brain is constantly mapping the external world like a GPS, even when we don’t know about it. This activity comes in the form of tiny electrical signals sent between neurons—specialized cells that communicate with one another to help us think, move, remember and feel. These signals often follow rhythmic patterns known as brain waves, such as slower theta waves and faster gamma waves, which help organize how the brain processes information.
Understanding how individual neurons respond to these rhythms is key to unlocking how the brain functions related to navigation in real time—and how it may be affected in disease.
A new study by Florida Atlantic University and collaborators from Erasmus Medical Center, Rotterdam, Netherlands, and the University of Amsterdam, Netherlands, has uncovered a surprising ability of brain cells in the hippocampus to process and encode and respond to information from multiple brain rhythms at once.
Huntington’s disease has long defied attempts to rescue suffering neurons. A new study in Cell Reports shows that transplanting healthy human glial progenitor cells into the brains of adult animal models of the disease not only slowed motor and cognitive decline but also extended lifespan. These findings shift our understanding of Huntington’s pathology and open a potential path to cell-based therapies in adults already showing symptoms.
“Glia are essential caretakers of neurons,” said Steve Goldman, MD, Ph.D., co-director of the University of Rochester Center for Translational Neuromedicine and lead author of the study.
“The restoration of healthy glial support—even after symptoms begin—could reset neuronal gene expression, stabilize synaptic function, and meaningfully delay disease progression. This study shifts the perspective on Huntington’s from a neuron-centric view to one that shows a critical role for glial pathology in driving synaptic dysfunction. It also tells us that the adult brain still has the capacity for repair when you target the right cells.”

Researchers at Karolinska Institutet and Lund University in Sweden have identified a new treatment strategy for neuroblastoma, an aggressive form of childhood cancer. By combining two antioxidant enzyme inhibitors, they have converted cancer cells in mice into healthy nerve cells.
The study, “Combined targeting of PRDX6 and GSTP1 as a potential differentiation strategy for neuroblastoma treatment,” is published in the journal Proceedings of the National Academy of Sciences.
Neuroblastoma is a type of childhood cancer that affects the nervous system and is the leading cause of cancer-related death in young children. Some patients have a good prognosis, but those with metastatic tumors often cannot be cured despite modern combinations of surgery, radiation, chemotherapy and immunotherapy.
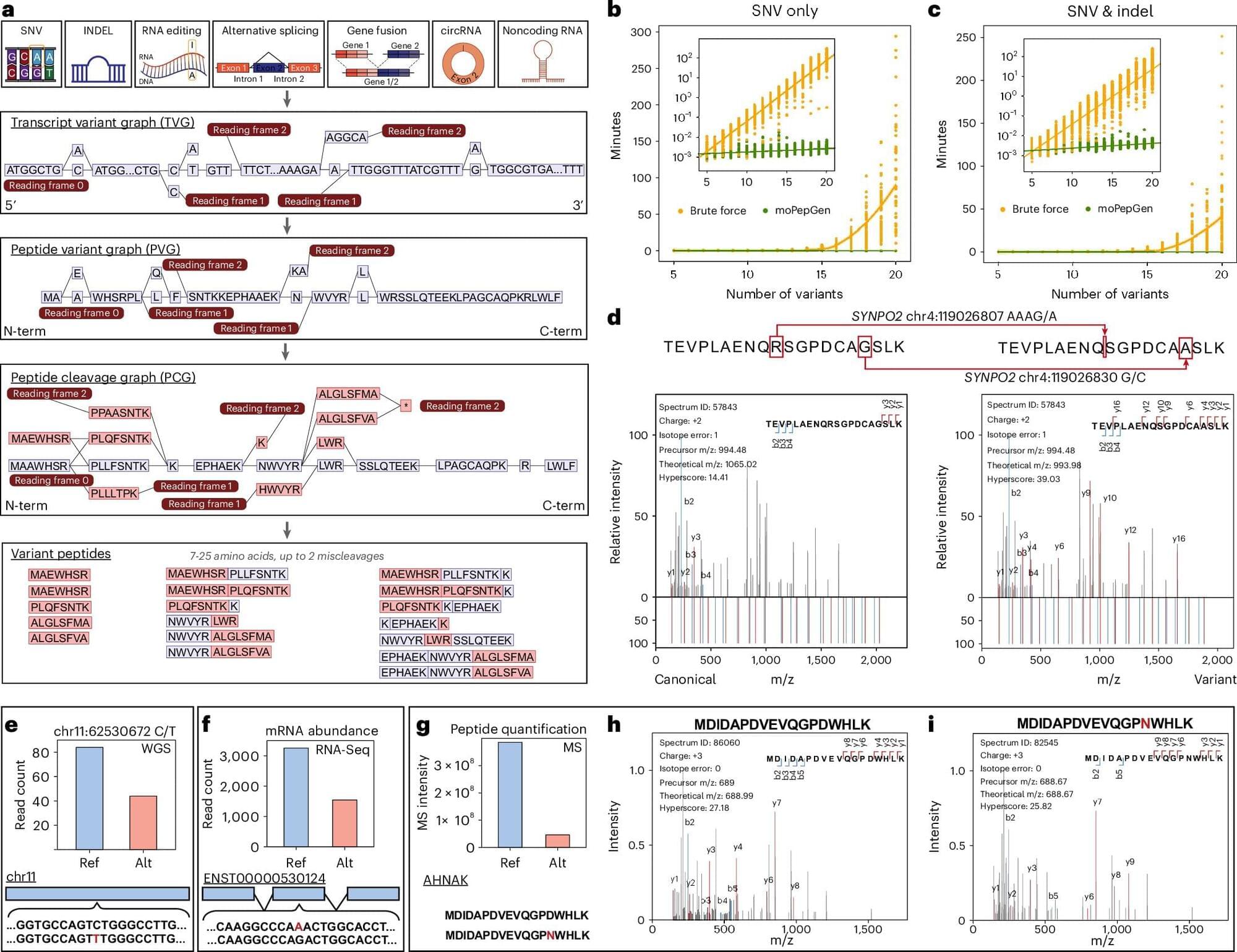
Scientists at UCLA and the University of Toronto have developed an advanced computational tool, called moPepGen, that helps identify previously invisible genetic mutations in proteins, unlocking new possibilities in cancer research and beyond.
The tool, described in Nature Biotechnology, will help understand how changes in our DNA affect proteins and ultimately contribute to cancer, neurodegenerative diseases, and other conditions. It provides a new way to create diagnostic tests and to find treatment targets previously invisible to researchers.
Proteogenomics combines the study of genomics and proteomics to provide a comprehensive molecular profile of diseases. However, a major challenge has been the inability to accurately detect variant peptides, limiting the ability to identify genetic mutations at the protein level. Existing proteomic tools often fail to capture the full diversity of protein variations.