How does Alzheimer’s target specific brain cells? Researchers are building a multiscale model of the hippocampus to map neuronal loss and cognitive decline.

New research has linked levels of vitamin D in midlife with toxic tangles of tau protein that accumulate in the brains of those with Alzheimer’s disease.
A statistical analysis of blood samples and brain scans from 793 adults showed that the more vitamin D in someone’s system in middle age, the lower the amount of tau protein tangles they tended to have years later.
The finding comes from an international team of researchers, and while it doesn’t prove direct cause and effect, it suggests an association that’s worth looking at.

A new Yale School of Medicine (YSM) study has uncovered surprising new details about how our eyes process what we see. When we look at something, our visual system breaks down different aspects of the scene—such as color, contrast, and motion—and processes those components separately. It’s called parallel visual processing and it’s what allows our brains to work out what we’re seeing so quickly.
This separation of information starts in the retina, and scientists have thought that separation is maintained as the information travels through the visual system. But in a study published in Neuron, researchers have found that information channels are more integrated than previously thought. This may help cells process weak visual signals, such as low-light conditions, the researchers say.
“We found that while different channels can deliver their own features, they’re also interconnected by underlying electrical circuitry,” says Yao Xue, Ph.D., a postdoctoral fellow in the department of ophthalmology and visual science at YSM and the study’s first author.
During intensive cancer treatment, children, adolescents and young adults are exposed to numerous toxicities and psychosocial stressors that can cause psychosocial distress and impair mental health. The maintenance or rapid recovery of mental health during and after exposure to significant stressors has been defined as resilience. To date, resilience research has focused primarily on cross-sectional assessment of specific, trait-like resilience factors and concepts in long-term survivors of childhood cancer, typically omitting the influence of context-specific biopsychosocial stressors and resilience dynamics throughout treatment. Little is known about outcome-based resilience and mental health resources in childhood cancer patients undergoing cancer treatment. In addition, specific instruments for age-appropriate assessment of resilience in childhood cancer patients are lacking. To address this gap, within the EU Horizon 2020-funded FORTEe project, we developed a novel self-report instrument for longitudinal assessment of resilience in children, adolescents, and young adults with cancer, featuring age-appropriate items tailored to their specific contexts.
An interdisciplinary team of psychologists, psychiatrists and pediatric oncologists developed an age-appropriate self-report instrument to assess resilience longitudinally in children, adolescents, and young adults undergoing cancer treatment. Following current resilience research frameworks, resilience is defined as the ratio of changes in mental health problems to stressor exposure. Accordingly, the measure comprises two domains: mental health problems (anxiety, depression, distress, fatigue) and stressor exposure (daily hassles, cancer-related stressors), with stressors rated for both frequency and intensity.
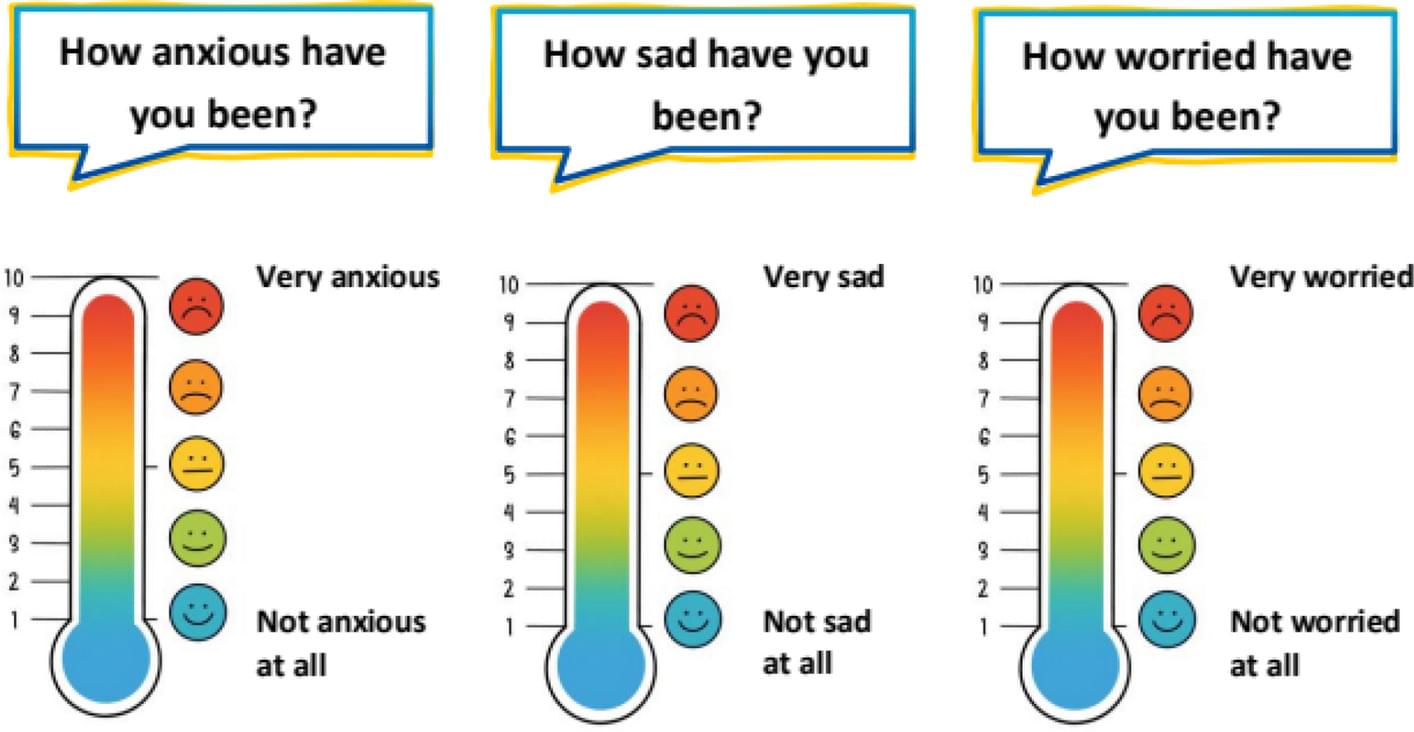
The Mainz Resilience Assessment in Childhood Cancer (MRAcc) consists of three age-specific versions (children 5–11 years, adolescents 12–17 years, young adults 18–21 years), each including the sections: ‘Emotions & Distress’, ‘Fatigue’, and ‘Situations & Experienced Stress’. It is available in German and English and uses either five-point-Likert scales or visual analogue scales presented as thermometers.
Carrying one or two copies of the APOE4 gene variant significantly increases the risk of developing Alzheimer’s, and a new study reveals how APOE4 can change neuron activity – potentially many years before symptoms such as memory loss start showing.
In young mice bred to have the APOE4 gene, researchers found specific neurons were smaller and more hyperactive in parts of the brain’s memory center, the hippocampus.
What’s more, they were able to identify a protein, Nell2, contributing to the disruption – and a potential pathway to reverse the damage in advanced cases.

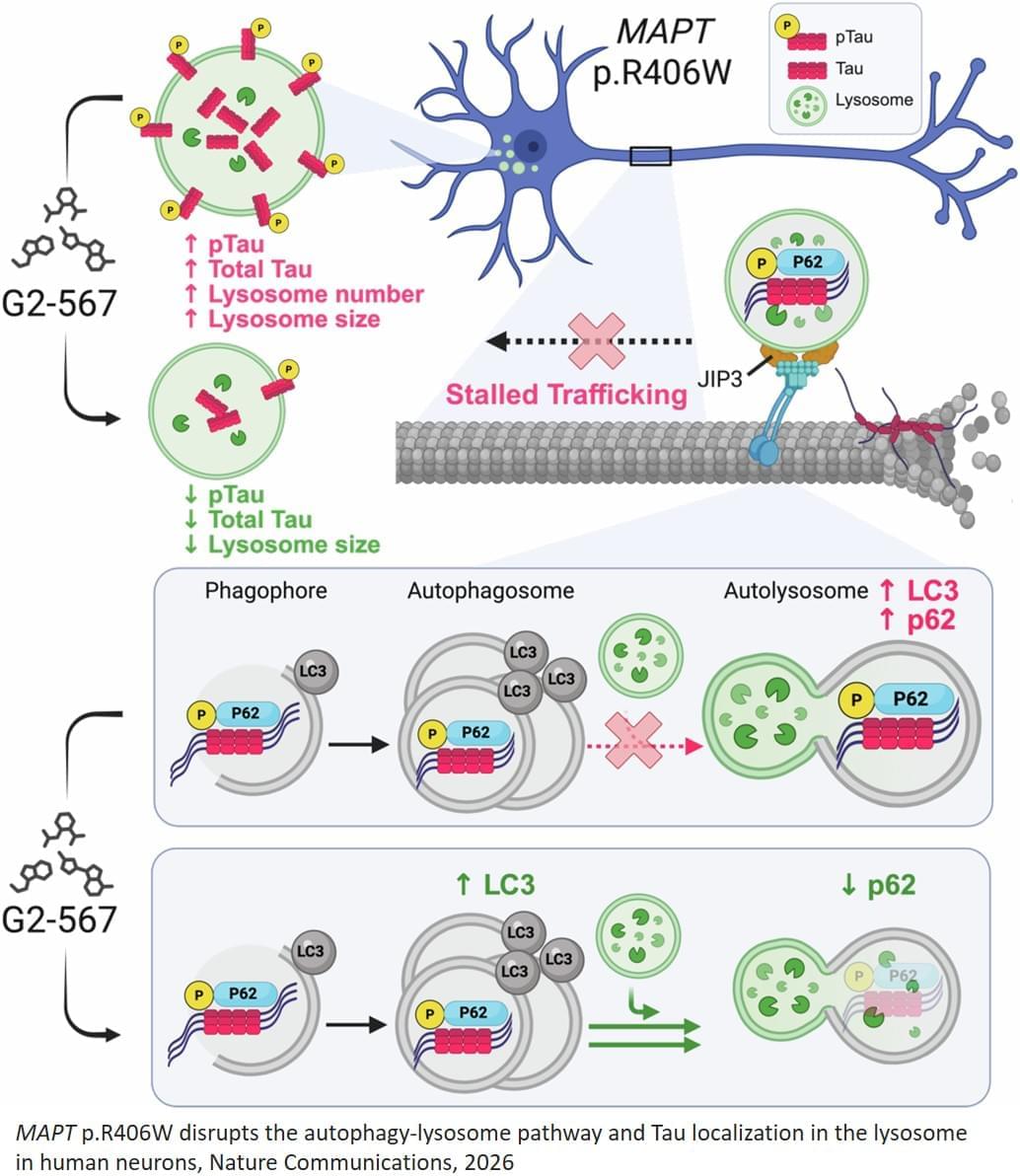
The researchers studied a specific mutation in a brain protein called tau that causes the protein to become misfolded and alter its normal function. In general, when tau proteins become misfolded, they build up inside neurons and contribute to various forms of dementia, including Alzheimer’s dementia and frontotemporal dementia, a neurodegenerative disease similar to Alzheimer’s that often strikes earlier — in middle age — and typically involves significant changes in personality and behavior that precede cognitive decline.
In this new study, the researchers studied neurons that had been reprogrammed from skin cells sampled from patients with frontotemporal dementia who carried the tau mutation. In the neurons, the mutated tau proteins caused waste-recycling centers called lysosomes, which are involved in autophagy, to become dysfunctional, allowing cellular waste to accumulate in the lysosomes, which may contribute to neuronal death. The researchers found that enhancing autophagy with an analog of the chemical compound G2 improved clearance of the garbage, reduced tau levels in the lysosomes and prevented cellular toxicity and death.
G2 was discovered in 2019 via screening experiments seeking drugs that could reduce the accumulation of an aggregation-prone protein in a C. elegans model of alpha-1-antitrypsin deficiency, which can cause severe liver disease. The compound was later shown to boost autophagy function in mammalian cell model systems.
The researchers also have shown that G2 can protect brain cells from death in cells modeling Huntington’s disease, a fatal inherited neurodegenerative disease caused by a genetic mutation present at birth. In the cellular model of Huntington’s disease, the compound prevented the buildup of a harmful RNA molecule. ScienceMission sciencenewshighlights.
New research adds to growing evidence that helping brain cells break down and eliminate their own cellular waste is a promising treatment strategy for a variety of neurodegenerative diseases. In lab experiments, the researchers found that exposure to a novel compound can clear a harmful protein from human neurons modeling frontotemporal dementia — a devastating and ultimately fatal condition — and prevent those neurons from dying.
The study is published in the journal Nature Communications.
Most of us have experienced that when our body is still and resting, the mind doesn’t stop. Instead, it takes off on its own journey of generating thoughts about our past, our plans, and the people around us, a process known as mind-wandering. While researchers have learned a lot about these kinds of thoughts, there aren’t many studies that explore how often our attention turns inward, toward sensations in our bodies, such as our breathing, heartbeat, or physical feelings.
This lesser-known side of our inner experience, called body-wandering, is what a recent study by a brain research team with collaborators from Denmark, Canada and Germany set out to explore.
To understand how the mind focuses on the physical self, researchers conducted a large-scale study with 536 participants who were asked to stay still in the MRI machine during a brain scan while looking at a cross on the screen above them.