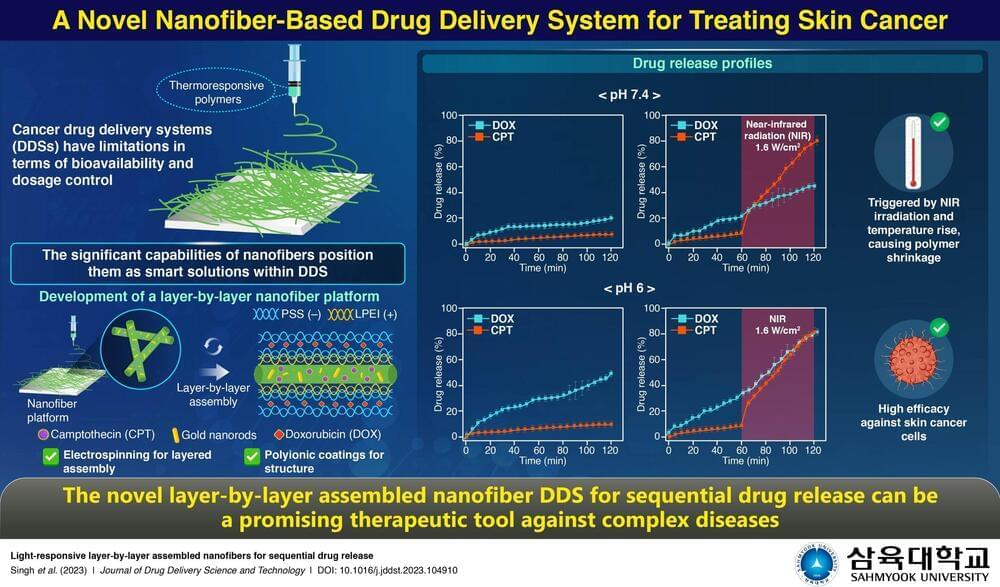
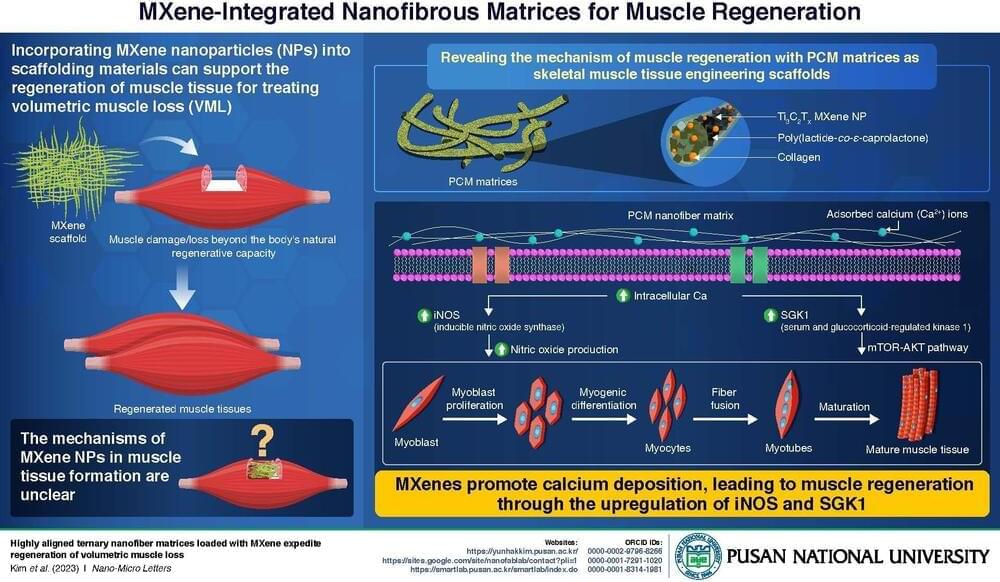
Treating complex diseases such as skin cancer often requires simultaneous administration of multiple anticancer drugs. The delivery of such life-saving therapeutic drugs has evolved with the rise of nanotechnology-based drug carriers. Nanoplatforms offer numerous advantages, including increased bioavailability, lowered dosages, and improved biodistribution.
Now a team of researchers, led by Professor Myoung-Hwan Park from Sahmyook University in South Korea, has developed a light-responsive nanofiber-based novel drug delivery system (DDS) targeting skin cancer. The DDS was studied in a detailed manner, beginning with its synthesis and characterization to its biocompatibility, drug release profile, and efficacy against skin cancer. These research findings are published in the Journal of Drug Delivery Science and Technology.
Explaining the motivation behind the present research, Dr. Park states, “Conventional drugs can be efficiently delivered in a controlled manner through nano-engineered platforms, and such an approach increases the overall effectiveness of the treatment. This approach improves outcomes in cancer drug therapy by ensuring precise delivery at optimal dosages.”