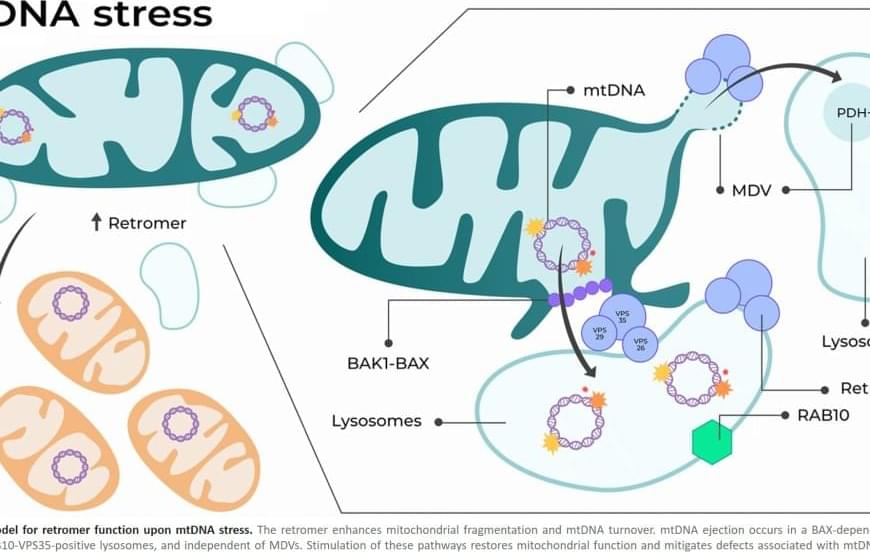
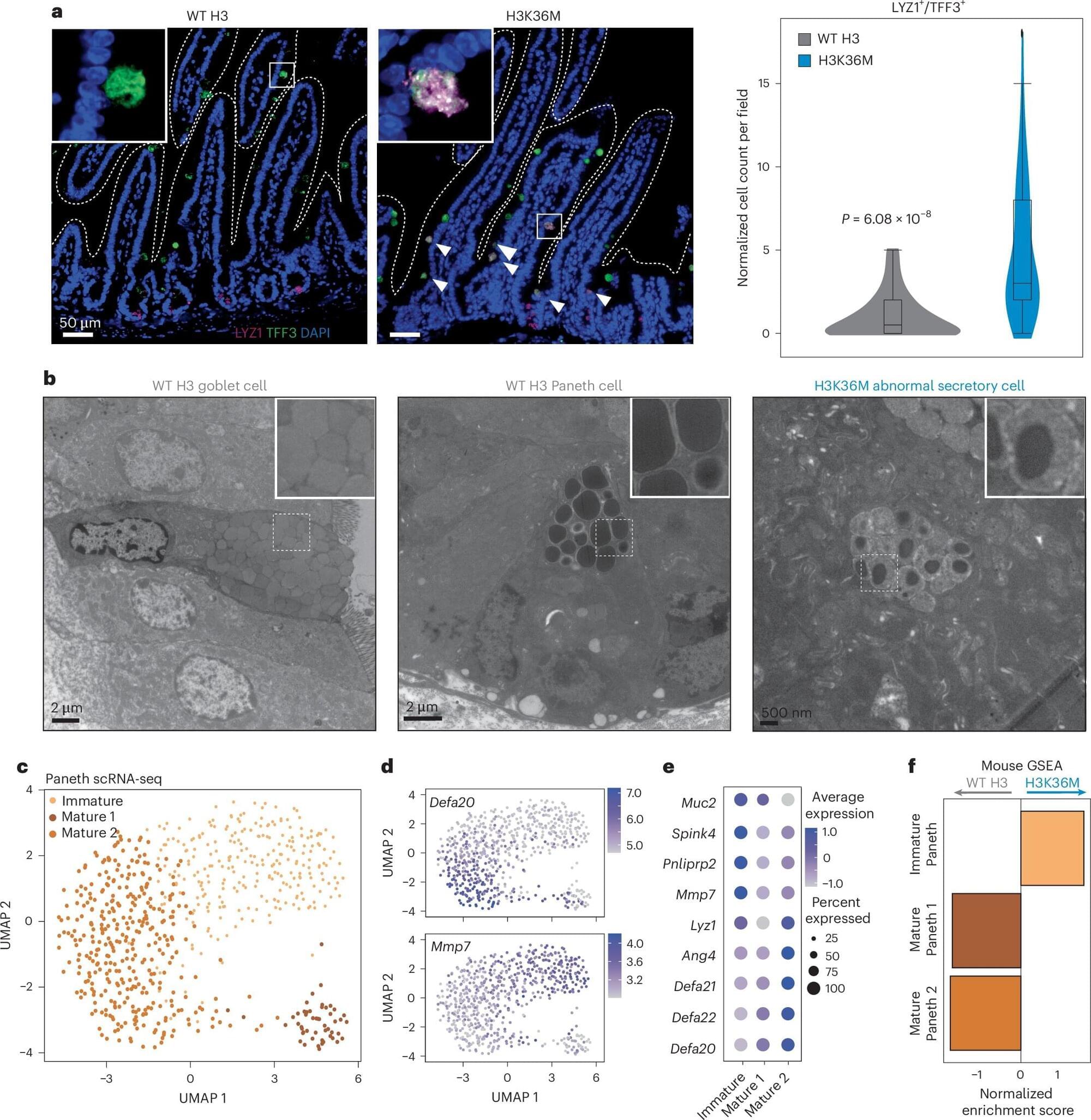
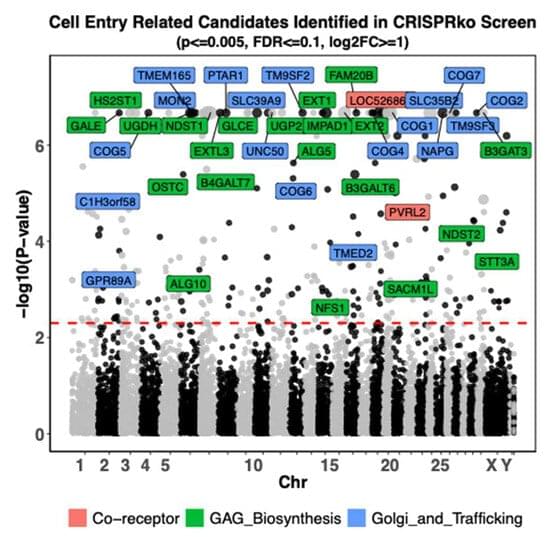
Researchers from the University of Colorado Cancer Center have solved a cellular mystery that may lead to better therapies for colorectal and other types of cancer.
Peter Dempsey, Ph.D., professor of pediatrics– developmental biology in the CU School of Medicine, and Justin Brumbaugh, Ph.D., assistant professor of molecular, cellular, and developmental biology at CU Boulder, recently published a paper in the journal Nature Cell Biology showing the importance of the H3K36 methylation process in regulating plasticity and regeneration in intestinal cells.
“The intestine has an enormous ability to regenerate itself after injury, and it does this through a model of dedifferentiation,” Dempsey explains. “The cells dedifferentiate back into a type of regenerative stem cell after injury, and those stem cells eventually recover the intestine and turn back to normal cells.”