Axolotls, hydra, and zebrafish can regenerate organs and bodies. Scientists say they may hold the key to healing, longevity, and scar-free recovery.


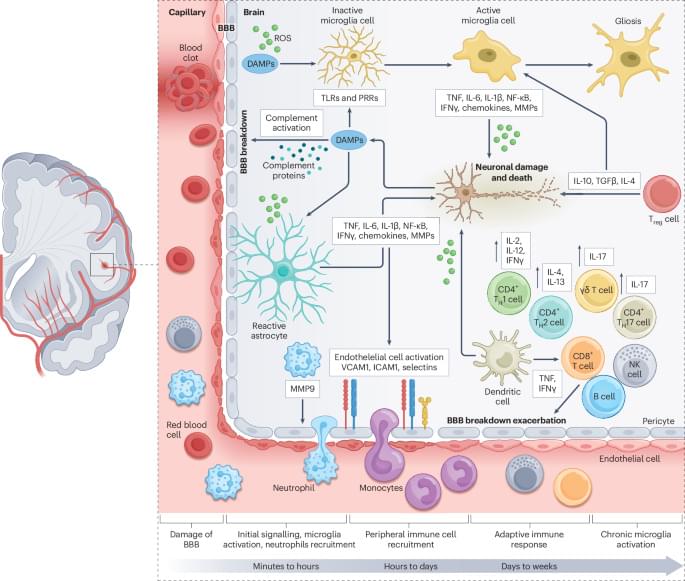
In this Review, Saef Izzy and colleagues examine the therapeutic potential of stem cells in stroke, with a focus on neural and mesenchymal stem cells. They explore how these stem cells interact with brain immune cells to modulate the inflammatory microenvironment, restore blood–brain barrier integrity and promote tissue repair following a stroke.
When disease begins forming inside the human body, something subtle happens long before symptoms appear. Individual molecules such as DNA, RNA, peptides, or proteins begin shifting in quantity or shape. Detecting these tiny molecular changes early could dramatically change how cancer, infections, and other conditions are diagnosed.
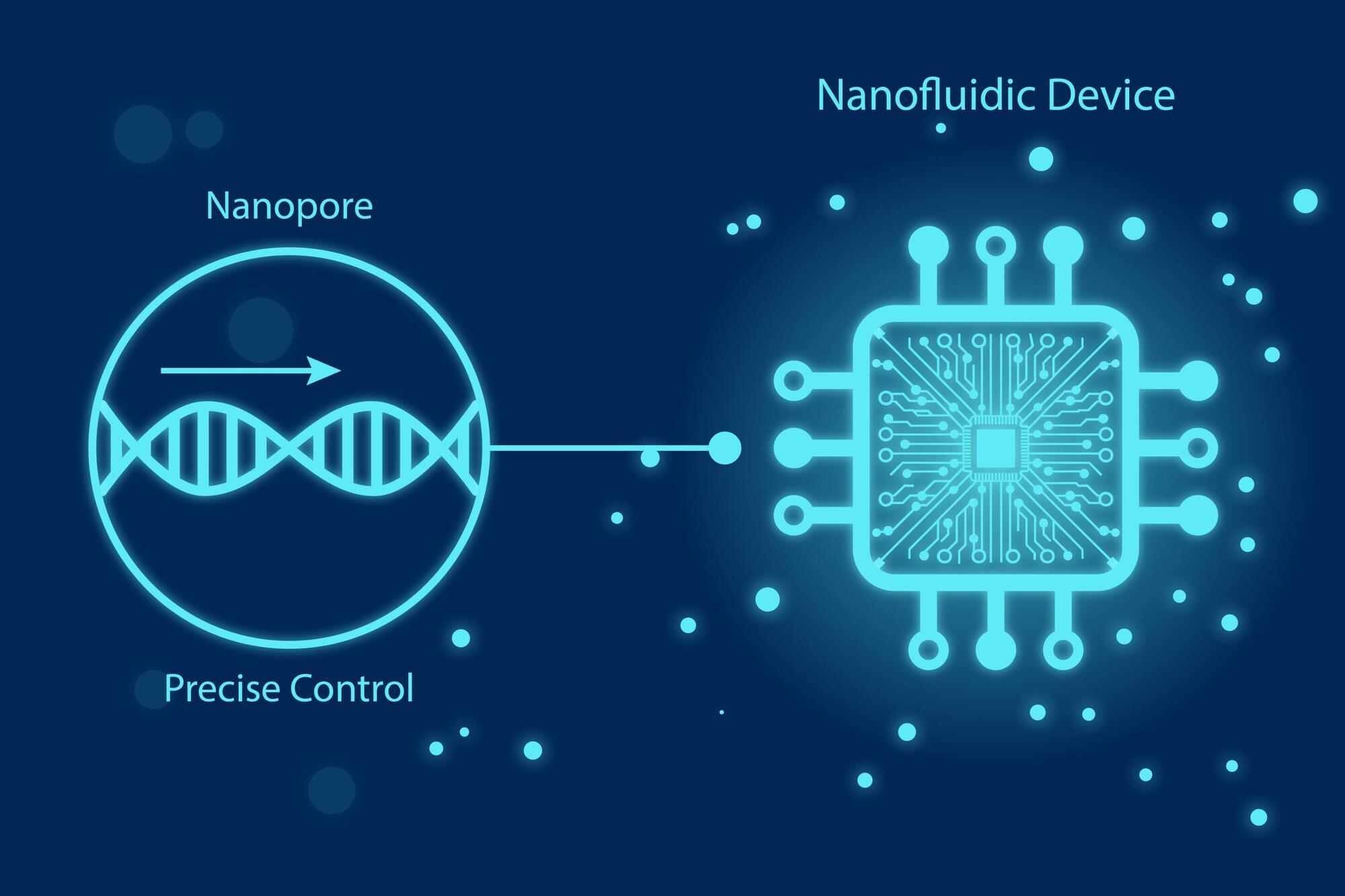
For years, scientists have dreamed of reading these changes one molecule at a time. Nanopores, nanometer-sized holes that detect single molecules by sensing changes in electric current, have brought that dream closer to reality, but nanopore sensing has hit several scientific walls.
Molecules rush through too fast to analyze. Signals drown in electrical noise. Proteins stick to pore surfaces and single pores simply cannot provide the scale required for real-world clinical use. So what would it take to make nanopore sensing fast, precise, and robust enough for society?
Studying the intricate molecular mechanisms that govern the assembly of the human nervous system has long been one of the most significant challenges in developmental biology and neuroscience. Researchers are continuously seeking a deeper understanding of how the human brain is built and what leads to various neurological disorders. Recent advancements in stem cell technology, particularly the ability to generate neural cells from pluripotent stem cells, coupled with the power of genome-editing tools like CRISPR-Cas9, are setting the stage for groundbreaking insights into human neurodevelopment and associated diseases. These technological innovations open new avenues for research that were previously thought to be unattainable.
The emergence of organoids and assembloids—miniature, simplified versions of brain tissue—has revolutionized the way scientists can model human development in vitro. Organoids replicate some of the complexity of human brain structures, allowing researchers to visualize developmental processes such as the specification, migration, and integration of neurons. This is particularly important for cortical interneurons, which migrate from the ventral forebrain to the dorsal forebrain during early brain development. These in vitro models provide an opportunity to study these intricate processes more closely and could lead to transformative discoveries in our understanding of brain diseases.
In a significant advancement outlined in recent research, scientists have developed a detailed protocol that marries pooled CRISPR-Cas9 screening with neural organoid and assembloid models. This innovative approach enables researchers to map hundreds of disease-related genes onto specific cellular pathways and critical aspects of human neural development. Such a strategy can significantly enhance our understanding of how various genes contribute to essential neuronal functions and the onset of neurological diseases, thereby paving the way for the development of novel therapeutic interventions.
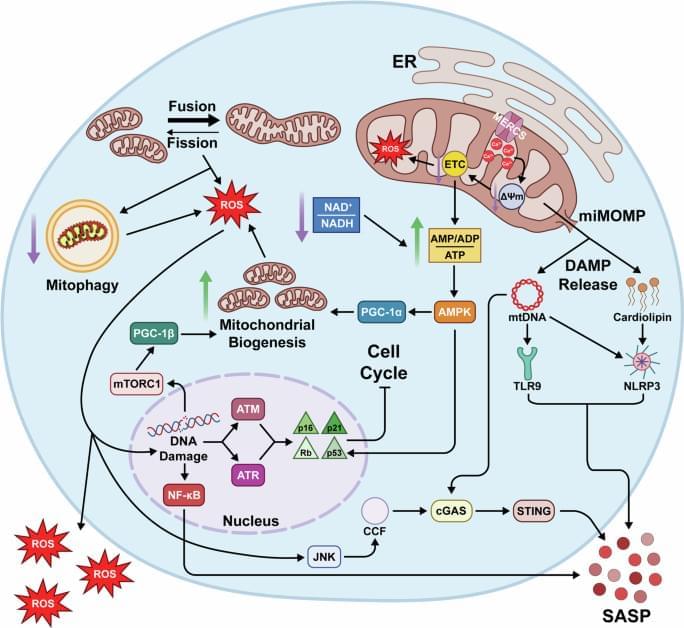
Hruby, A.J., Higuchi-Sanabria, R. Mitochondrial dysfunction in cellular senescence: a bridge to neurodegenerative disease. npj Aging 11, 99 (2025). https://doi.org/10.1038/s41514-025-00291-4
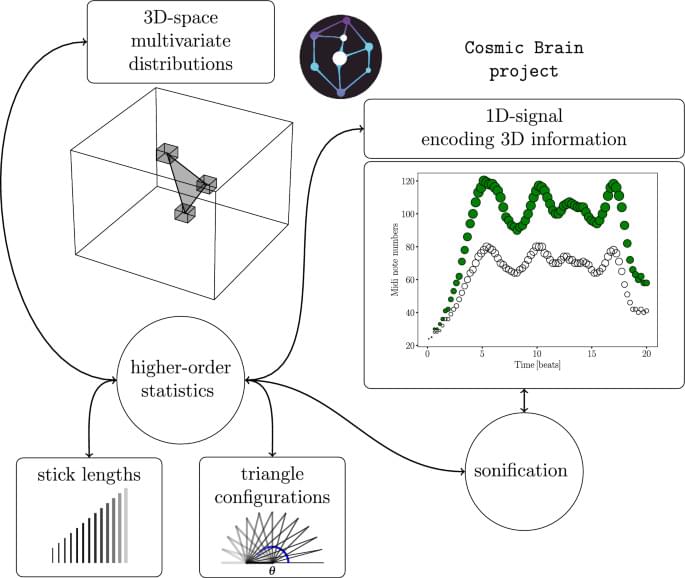
Scientific Reports volume 15, Article number: 42,309 (2025) Cite this article.


Researchers have found that they could use highly insulating aluminum-coated polymer film to improve the performance of flexible electronics and medical sensors.
Currently, the aluminum-coated polymer film is used to shield satellites from temperature extremes.
Researchers at Empa have succeeded in making the material even more resistant by implementing an ultra-thin intermediate layer.
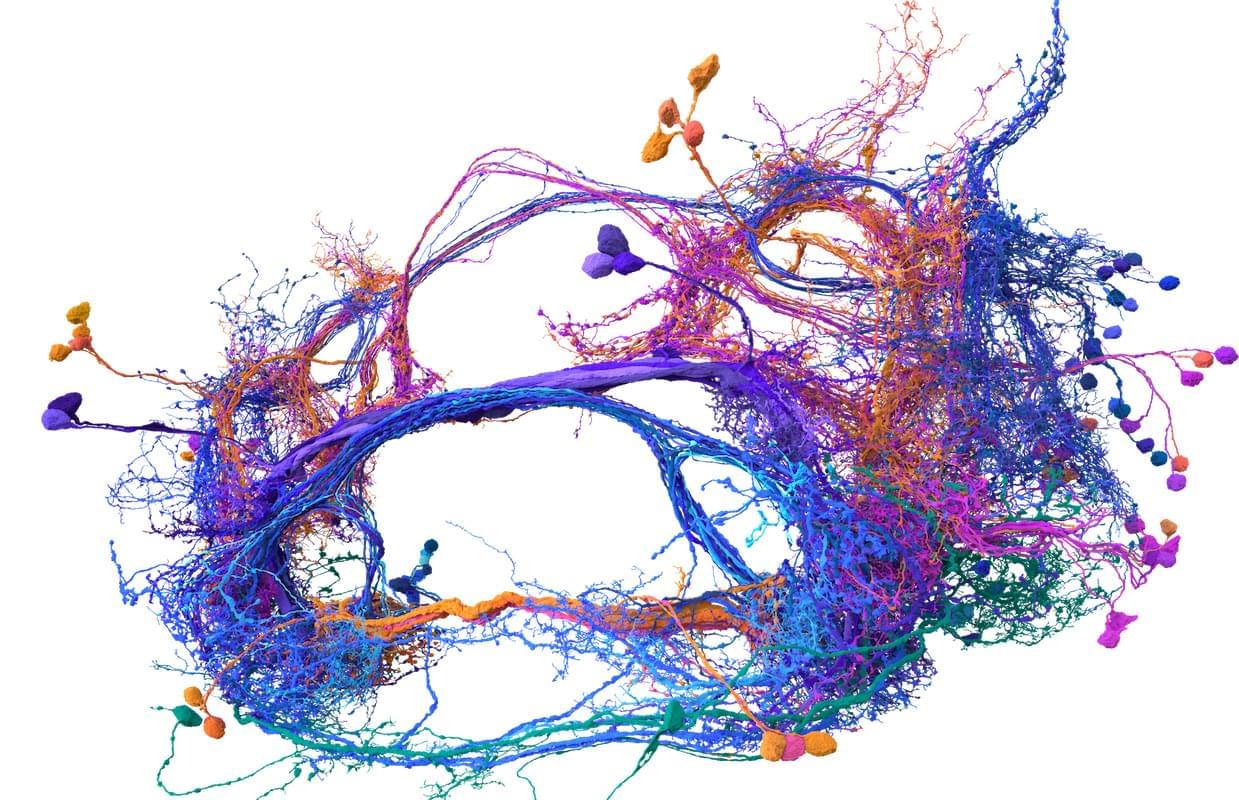
On a Sunday evening earlier this month, a Stanford professor held a salon at her home near the university’s campus. The main topic for the event was “synthesizing consciousness through neuroscience,” and the home filled with dozens of people, including artificial intelligence researchers, doctors, neuroscientists, philosophers and a former monk, eager to discuss the current collision between new AI and biological tools and how we might identify the arrival of a digital consciousness.
The opening speaker for the salon was Sebastian Seung, and this made a lot of sense. Seung, a neuroscience and computer science professor at Princeton University, has spent much of the last year enjoying the afterglow of his (and others’) breakthrough research describing the inner workings of the fly brain. Seung, you see, helped create the first complete wiring diagram of a fly brain and its 140,000 neurons and 55 million synapses. (Nature put out a special issue last October to document the achievement and its implications.) This diagram, known as a connectome, took more than a decade to finish and stands as the most detailed look at the most complex whole brain ever produced.
Meet Memazing.
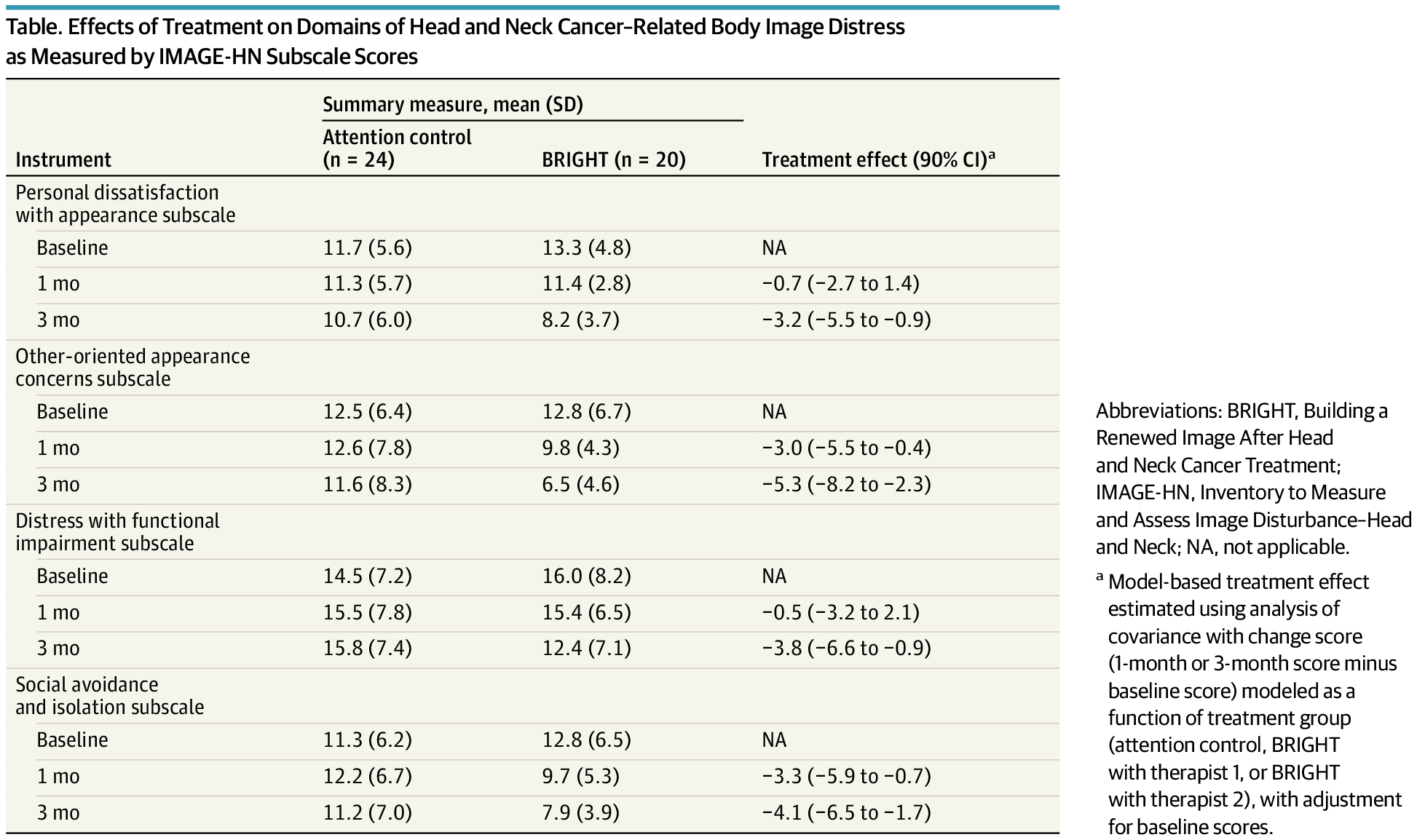
The BRIGHT program, a brief cognitive behavioral treatment, effectively reduced body image distress across multiple domains in head and neck cancer survivors.
This secondary analysis of a randomized clinical trial evaluates whether a brief, tailored cognitive behavioral treatment program is effective across multiple domains of head and neck cancer–related body image distress.