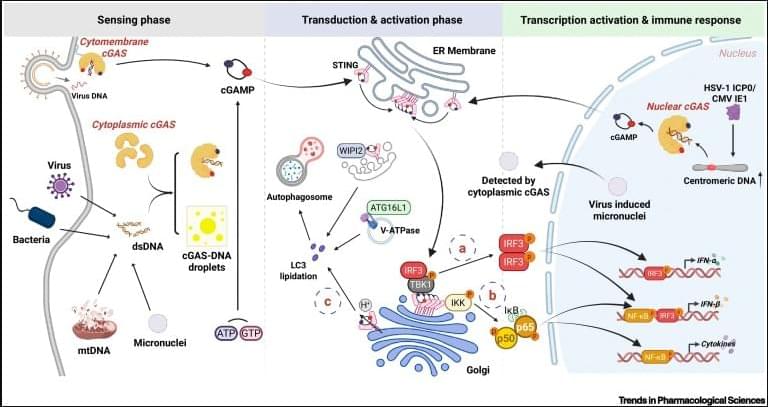
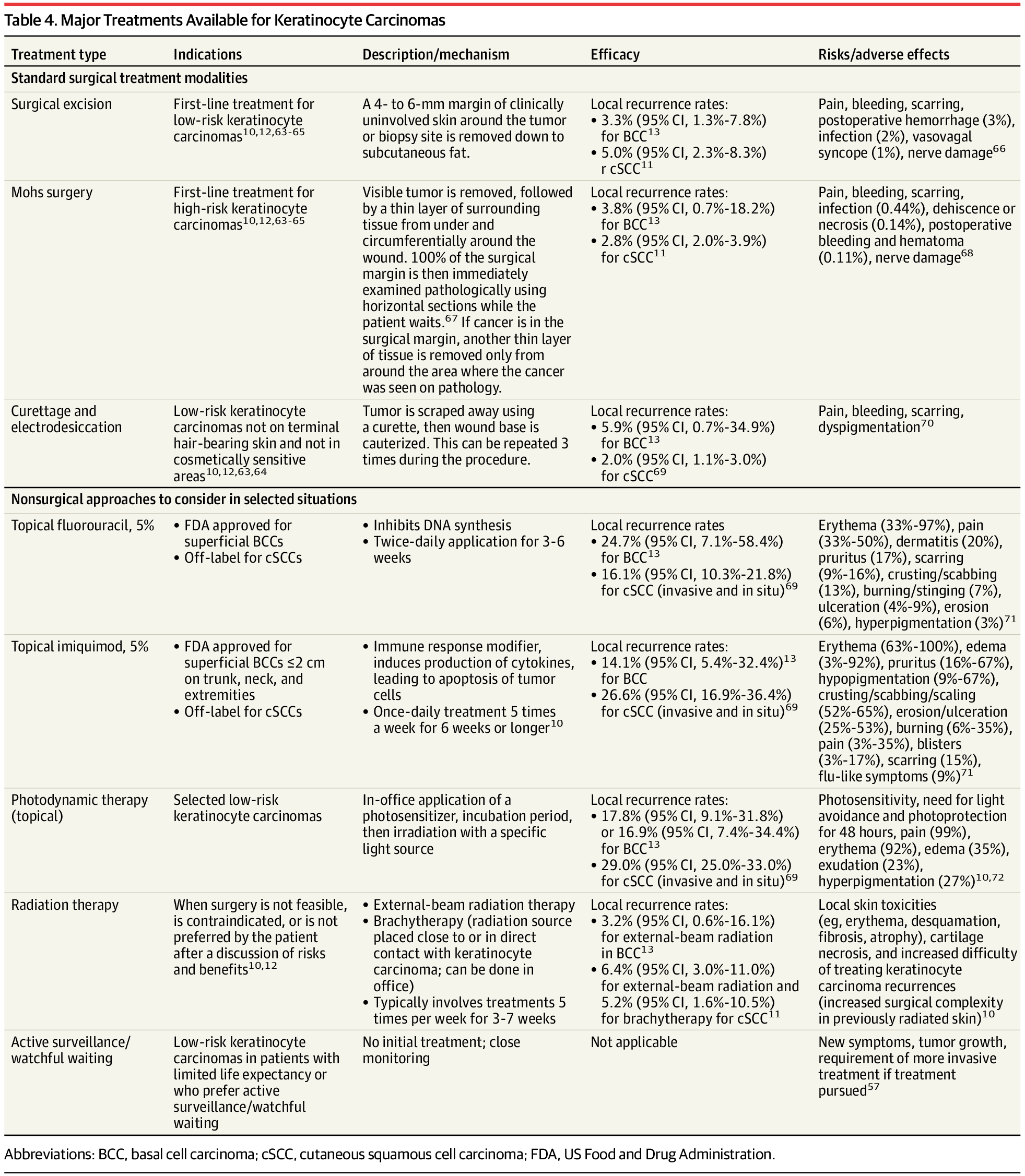
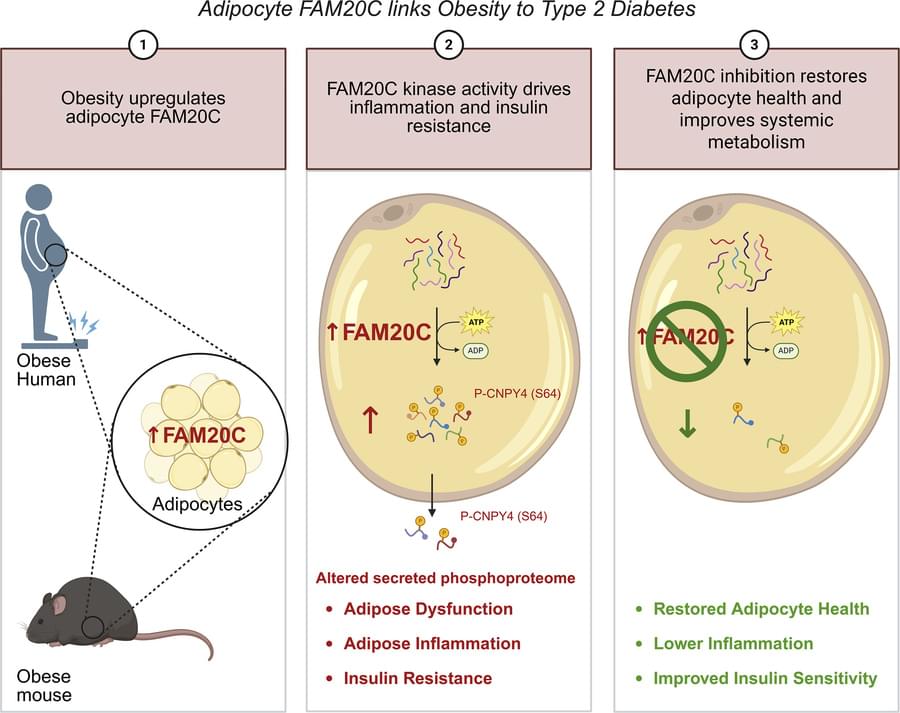
A regular heartbeat is essential to vertebrate life. In the mature heart, this function is driven by an anatomically localized pacemaker. By contrast, pacemaking capability is broadly distributed in the early embryonic heart1–3, raising the question of how tissue-scale activity is first established and then maintained during embryonic development. The initial transition of the heart from silent to beating has never been characterized at the timescale of individual electrical events, and the structure in space and time of the early heartbeats remains poorly understood. Using all-optical electrophysiology, we captured the very first heartbeat of a zebrafish and analysed the development of cardiac excitability and conduction around this singular event. The first few beats appeared suddenly, had irregular interbeat intervals, propagated coherently across the primordial heart and emanated from loci that varied between animals and over time. The bioelectrical dynamics were well described by a noisy saddle-node on invariant circle bifurcation with action potential upstroke driven by CaV1.2. Our work shows how gradual and largely asynchronous development of single-cell bioelectrical properties produces a stereotyped and robust tissue-scale transition from quiescence to coordinated beating.
© 2023. The Author(s), under exclusive licence to Springer Nature Limited.