Ovarian cancer kills more women than any other gynecological cancer. Most patients receive their diagnosis only after the disease spreads throughout the abdomen. Until now, scientists have never fully understood why this cancer advances so fast.
A new study led by Nagoya University explains why. Published in Science Advances, the study shows that cancer cells recruit help from protective mesothelial cells that normally line the abdominal cavity. Mesothelial cells lead the invasion and cancer cells follow the pathways they create. These hybrid cell clusters resist chemotherapy better than cancer alone.
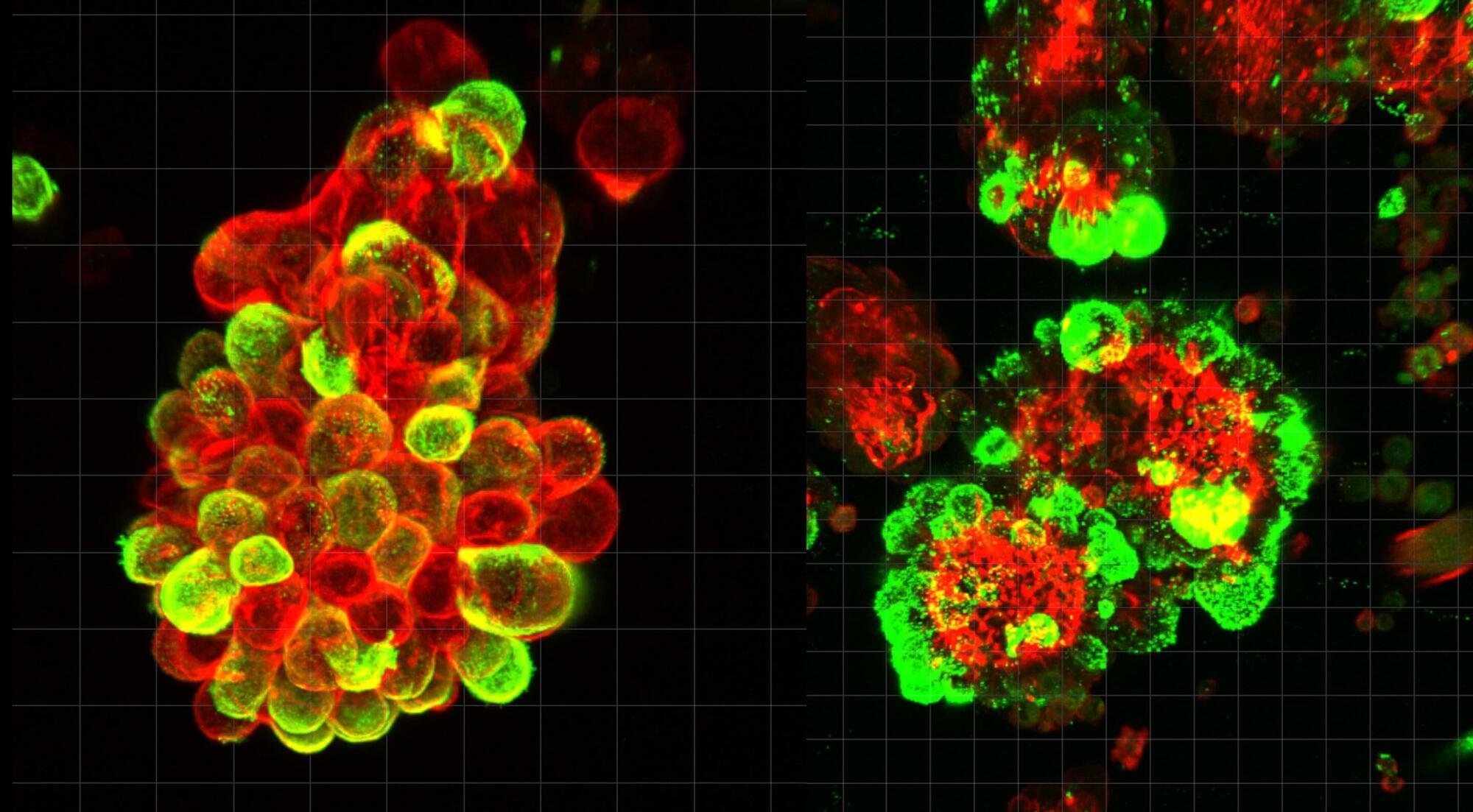
Researchers examined abdominal fluid from ovarian cancer patients and found something unexpected. Cancer cells do not float alone in the abdominal cavity. Instead, they often grab onto mesothelial cells and form hybrid spheres. About 60% of all cancer spheres contain these recruited mesothelial cells. The cancer cells release a protein called TGF-β1 that transforms the mesothelial cells and causes them to develop spike-like structures that cut through tissue.